Biosecurity
thumbnail
Livestock Management
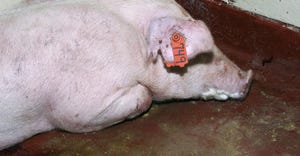
Allerson named 2024 Allen D. Leman Science in Practice Award winnerAllerson named 2024 Allen D. Leman Science in Practice Award winner
Holden Farms swine veterinarian renowned for his expertise in disease control, health and production.
Subscribe to Our Newsletters
National Hog Farmer is the source for hog production, management and market news













.jpg?width=300&auto=webp&quality=80&disable=upscale)