Senecavirus A impacting neonatal mortality
September 28, 2015

Neonatal piglet mortality is caused by different factors, including non-infectious and infectious conditions. Among the infectious conditions present in the United States, pathogens associated with substantial increases in mortality include porcine reproductive and respiratory syndrome virus and a few enteric pathogens as porcine epidemic diarrhea virus, transmissible gastroenteritis virus, rotavirus and E.coli. There might be another pathogen collaborating with neonatal mortality, as suggested recently in the Brazilian and U.S. swine industries 2,3.
Senecavirus A (formerly known as Seneca Valley virus) is a Picornavirus (small RNA virus), first identified as a cell culture media contaminant4,5, and proposed to be a non-pathogenic virus that has oncolytic properties and thus useful for cancer therapy in humans. However, Senecavirus A has also been frequently identified in pigs with idiopathic vesicular disease and therefore proposed as causative agent of vesicular disease in pigs6,7.
Recently, case reports from Brazil and the Midwestern U.S. have identified Senecavirus A in cases of neonatal mortality. This column summarizes the current knowledge about Senecavirus A and the recently described neonatal losses syndrome.
The neonatal losses syndrome affected pigs of 0-7 days of age. Piglets of 0-3 days of age had a high fatality rate (40-80%). Piglets from 4-7 days of age showed a moderate fatality rate (0-30%). Affected sow farms had a sudden onset (increase) of neonatal mortality, and recovered to baseline mortality levels within four to 10 days. Because of those characteristics, this syndrome has been referenced as epidemic transient neonatal mortality3.
Extensive pathological evaluation of affected piglets from several herds did not find common macroscopic (gross lesions) or microscopic (i.e. histopathology) lesions that could explain the cause of piglet mortality. The common finding of such herds is the detection of large amounts of Senecavirus A from multiple piglet tissues, including brain, blood and lymphoid tissues, indicating a widespread infection.
Good news is that breeding herds that experienced ETNL did not re-break after the initial outbreak (one year now), meaning that assuming Senecavirus A is the etiological agent, it has a high transmission rate (infects the whole herd within a few days) and elicits a fast, strong, long lasting protective immunity.
ETNL and idiopathic vesicular disease
Many ETNL cases have also reported idiopathic vesicular disease in sows3. Briefly, it has been proposed the term “idiopathic vesicular disease” for vesicular disease cases proved negative to the four classical viruses (FMDv, VSv, SVDv and VESv). These four vesicular diseases are all foreign animal diseases, in that they are not currently present in the U.S. national swine herd. IVD was described in Oceania, Europe, North America and, more recently, in Brazil.
Senecavirus A has been identified at high infection loads in several independent IVD cases, including exhibit pigs as well as commercial pigs, indicating that Senecavirus A is likely a causative agent of vesicular disease in pigs.
ETNL distribution
The ETNL syndrome in Brazil started in September 2014, and moved quickly to all major commercial-pig producing states (Figure 1). At a recent panel between leaders of the Brazilian industry it has been agreed that at least 70% of the industry has experienced ETNL from September 2014 to present date.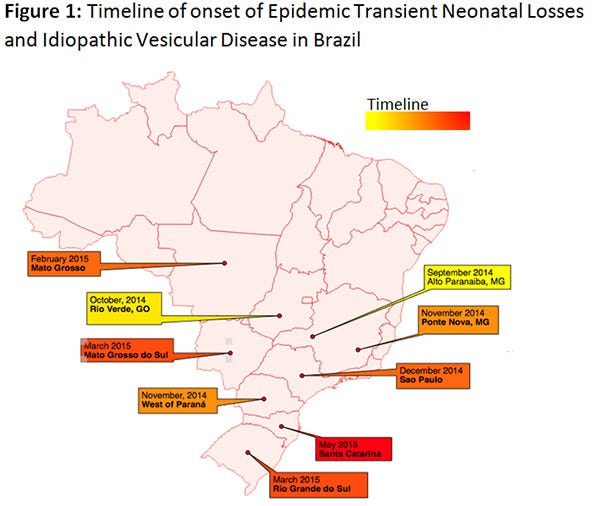
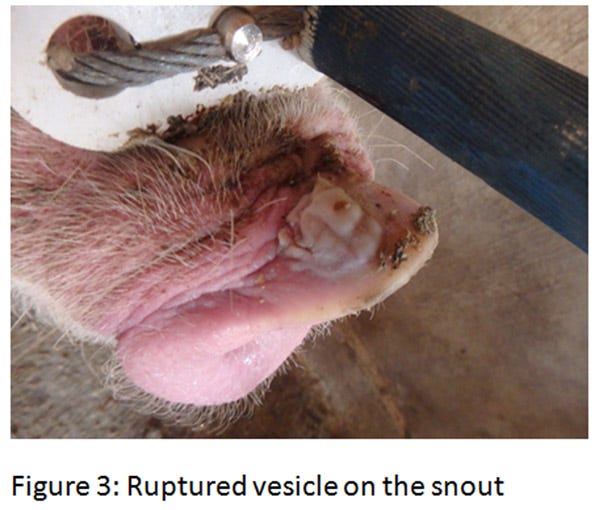
The ETNL syndrome in the United States was reported in approximately 40 breeding herds in the Midwest. Similar to Brazil’s description, the increase in neonatal mortality lasts for about a week and suddenly returns to baseline. Upon further investigation, vesicles on the snout or coronary bands are sometimes discovered on some sows (see Figures 2-4)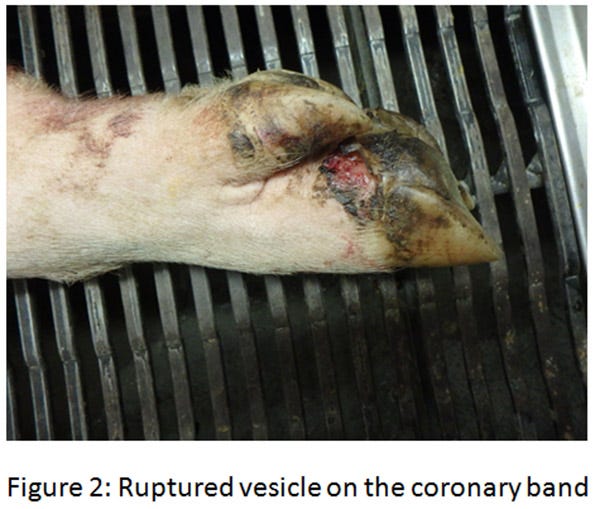
 Senecavirus A is circulating at low prevalence in the United States. A recent retrospective study conducted by Iowa State University and University of Minnesota Veterinary Diagnostic Laboratories (funded by the Swine Health Information Center) showed a low prevalence (n=five cases) of Senecavirus A detection by PCR in oral fluids (441 cases, 2,033 samples, 25 states) received for routine tests for other pathogens (e.g. PRRSv, influenza). Each case was from a different state (Arkansas, Iowa, Indiana, Illinois and Minnesota).
Senecavirus A is circulating at low prevalence in the United States. A recent retrospective study conducted by Iowa State University and University of Minnesota Veterinary Diagnostic Laboratories (funded by the Swine Health Information Center) showed a low prevalence (n=five cases) of Senecavirus A detection by PCR in oral fluids (441 cases, 2,033 samples, 25 states) received for routine tests for other pathogens (e.g. PRRSv, influenza). Each case was from a different state (Arkansas, Iowa, Indiana, Illinois and Minnesota).
It is also important to highlight that viruses isolated from recent ETNL cases are genetically similar between themselves (between and within Brazilian and U.S. cases). However, contemporary Senecavirus A isolates are significantly different from most historical isolates from the United States and Canada (Jianqiang, personal information).
Comparisons between Senecaviruses were made between the whole genome sequences, and also between the VP1-region, which is a more variable segment, thought to be associated with neutralizing antibodies.
Prevention
At this point it is not completely understood how Senecavirus A and/or ETNL disseminates between pig populations. There were a few affected breeding herds that were relatively well isolated, without recent history of animal introduction (i.e.: closed for at least 180 days), which suggests that ETNL were transmitted by routes other than pig movement.
Senecavirus A is another example that the livestock industry must be prepared for pathogen and disease emergence. Strict biosecurity measures must be implemented at all times, regardless of the farm health status. In other words, herds that are “already” positive for PRRSv or Mycoplasma or influenza are “still” free of many other infectious agents and should implement all applicable biosecurity practices 7/24/365.
What should be done when vesicular disease is suspected?
When vesicular disease is suspected (e.g.: lameness, snout and/or coronary bands lesions), the farm veterinarian and state/federal officials should be contacted immediately (before any further pig movements). State and federal officials will determine the next course of actions.
Because of the similarities to vesicular diseases that are foreign animal diseases, we need to be diligent to test these cases and make sure they are associated with Senecavirus A and not one of these other vesicular diseases.
1. Amass SF, Schneider JL, Miller CA, Shawky SA, Stevenson GW, Woodruff ME. Idiopathic vesicular disease in a swine herd in Indiana. J Swine Health Prod. 2004;12(4):192-196.
2. Linhares D, Rademacher C, Vannucci F, Barcellos D. Seneca-associated diseases - clinical presentation and epidemiological investigation, at: Allen D Leman Swine Conference 2015; St Paul, MN.
3. Vannucci FA, Linhares DC, Barcellos DE, Lam HC, Collins J, Marthaler D. Identification and Complete Genome of Seneca Valley Virus in Vesicular Fluid and Sera of Pigs Affected with Idiopathic Vesicular Disease, Brazil. Transbound Emerg Dis. 2015.
4. Hales LM, Knowles NJ, Reddy PS, Xu L, Hay C, Hallenbeck PL. Complete genome sequence analysis of Seneca Valley virus-001, a novel oncolytic picornavirus. J Gen Virol. 2008;89(Pt 5):1265-1275.
5. Reddy PS, Burroughs KD, Hales LM, et al. Seneca Valley virus, a systemically deliverable oncolytic picornavirus, and the treatment of neuroendocrine cancers. J Natl Cancer Inst. 2007;99(21):1623-1633.
6. Leme RA, Zotti E, Alcântara BK, et al. Senecavirus A: An Emerging Vesicular Infection in Brazilian Pig Herds. Transbound Emerg Dis. 2015.
7. Singh K, Corner S, Clark S, Scherba G, Fredrickson R. Seneca Valley Virus and Vesicular Lesions in a Pig with Idiopathic Vesicular Disease. Journal of Veterinary Science & Technology. 2012;3(123):1-3.
You May Also Like



