Approach to COVID-19 similar to battling swine pathogens
Herd immunity is a key concept for both swine and public health.
April 21, 2020

One of the most important things to understand is the concept of herd immunity. Individual animals can become immune by recovering from an earlier infection or through vaccination. Some animals cannot become immune due to their age, stress (weaning, environmental conditions), co-infections and for this group herd immunity is a crucial method of protection.
Once a certain threshold of the population is immune, herd immunity gradually eliminates a disease from a population. The term "herd immunity" implies that it must have been developed in livestock but actually it was a term first developed in the 1930s to describe a phenomena observed after a significant number of children had become infected and immune to measles, the number of new infections temporarily decreased, including among susceptible children. It was soon realized that not only immunity from infections, but also from vaccination could also provide "herd immunity."
Herd Immunity and R0
Herd immunity is used to describe the threshold proportion of immune animals that should lead to a decline in incidence of infection attributable to immunity acquired via infection and/or vaccination. The risk of infection to susceptible individuals in the herd is reduced because of the presence, proximity and number of immune individuals present in the population (Figure 1).
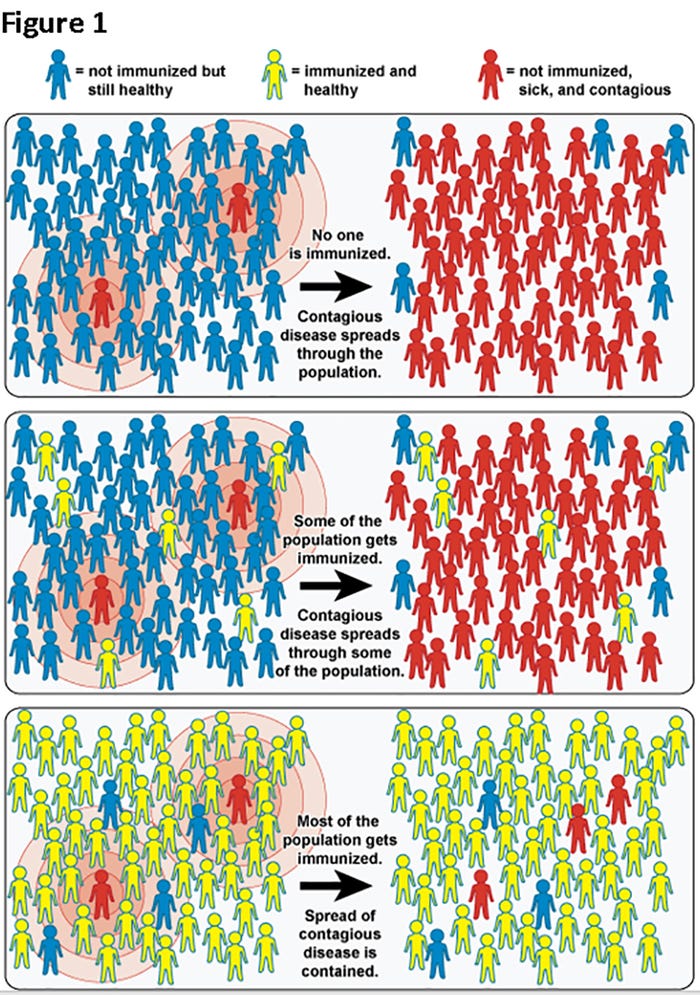
As can be seen in top panel of Figure 1, 100% of the individuals are susceptible so when a sick individual enters the group, everybody is susceptible and can become infected. In the middle panel, there are few individuals who are immunized but not enough to stop the spread of the disease (Figure 1, middle panel). In the bottom panel, there are enough immunized individuals to "protect" some of the susceptible individuals because even when virus infects "immunized" individuals it can't replicate and can't be shed to the susceptible individuals (Figure 1, bottom panel).
With regard to swine vaccination, one of the key factors to consider is that the level of protection conferred by most vaccines does not fully protect the herd against infection. In this case, there may be a "critical threshold," that is, the fraction of the population that need to be immunized and develop an immune response (according to the level of immunization conferred by the vaccine) to mitigate a disease outbreak (Figure 2).
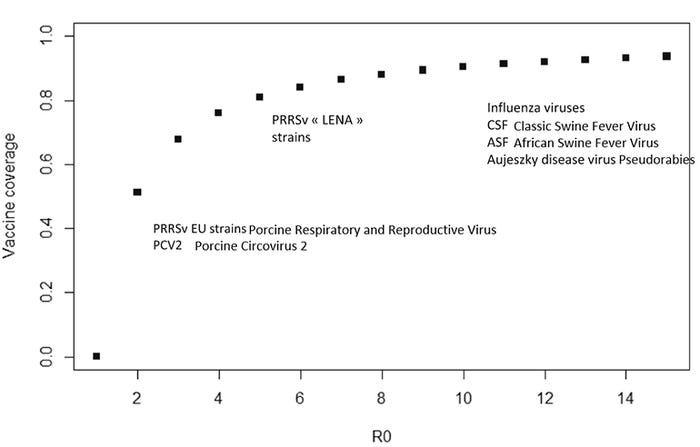
As illustrated in Figure 2, on the Y-axis, the percentage of vaccine coverage varies by virus. Vaccine coverage (aka as threshold of protection) is the percentage of animals that are immune following vaccination. In the case of porcine circovirus 2 and European strain of porcine respiratory and reproductive virus, the "herd immunity" (partial protection for the susceptible animals) can develop when only 50% of the animals develop immunity (vaccine coverage) and when the herd reaches 80% vaccine coverage, the herd should theoretically have pathogen eradication (Rose and Andraud 2017).
In contrast, the highly contagious foreign animal diseases, including classic swine fever and African swine fever viruses require vaccine coverage of almost 100%. The protection of a herd from vaccines results from a variety of factors: the vaccine can reduce the susceptibility of pigs to the infection; it can decrease pathogen disease severity once the pig is infected, or accelerate the elimination of the pathogen by reducing pathogen replication or duration of shedding. The pathogen factor that influences the level of vaccine coverage is R0 (Reproduction rate of virus infection) (Figure 2 X-axis).
The R0 (pronounced "r naught") tell us how easily a virus spreads from animal to animal (Figure 3). In Figure 3, we illustrate the R0=3 which means that one individual can infect three other individuals. For examples, PCV2 and European PRRSV have been estimated to have an R0 of 2 to 5 while some strains of ASF have been estimated to have an R0 as high as 18: one infected animal could infect 18 other pigs (Figure 2 X-axis) (Schulz et al 2017).
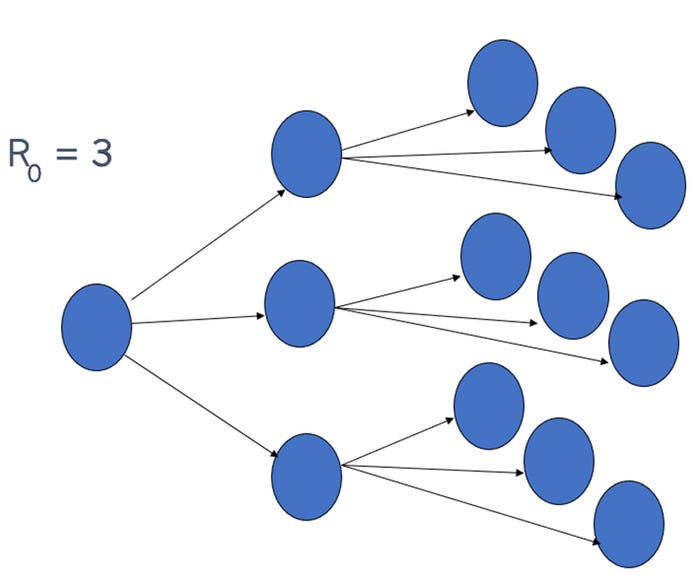
Herd immunity following vaccination varies by pathogen and formulation. For example, PCV2 vaccine herd immunity is generally high. In contrast, PRRSV vaccines only provide a limited protection from an epidemiological point of view, and would be generally called a "leaky vaccine" (Rose and Andraud 2017). Stocking density is a management factor that will have a huge effect on disease spread as the higher density of "susceptible animals" will result in rapid disease development.
Vaccination and exposure approaches and herd immunity
Vaccine responses vary by the age of the host, host immune response, pathogen load and the antigenic variability of the pathogen. "Single dose" vaccines are common for endemic diseases like PCV2 and Mycoplasma hyopneumoniae. These vaccines induce immunity and the "booster" is actually the exposure to the pathogen in the environment. These pathogens are relatively stable antigenically.
Pathogens that require booster vaccine usually are in lower levels in the environment and/or the host immune response needs additional immune stimulation, requiring vaccine formulations containing an adjuvant. Finally, there are pathogens where antigenic variability is ongoing and/or host response to commercial vaccines is poor (i.e. PRRSV and swine influenza virus). The approaches used in these situations may include autogenous vaccines. In the case of PRRSV and porcine epidemic diarrhea virus often controlled exposure by natural infection is necessary to develop an adequate immune response in replacement gilts that will result in herd immunity.
COVID19 and herd immunity
With COVID-19 and the human population, all of the principles we have discussed above apply. As we are seeing with COVID-19, a 100% susceptible population is giving us the situation in the top panel of Figure 1.
As we socially isolate, we are decreasing the density so the R0 of 2 to 3 can be minimized (Table 1). We are attempting to keep the R0 low until there is a sufficient proportion of the population immune to provide herd immunity. The problem is that exposure is the only mechanism to induce immunity until a vaccine is available. Many "cold viruses" have no or minor symptoms and exposure is the way we develop herd immunity. However, with a virus as deadly as COVID-19, vaccination is the only way to safely attain herd immunity. The R0 for the common human childhood diseases vary dramatically (Table 1). Prior to the COVID-19 pandemic, one of the biggest public health issues in the United States was measles. As seen in Table 1, the reason for the concern with the decrease use of measles vaccines is that measles has a high R0 of 12 to 18, requiring vaccine coverage of 83% to 94% to achieve herd immunity. Decreasing vaccination then decreases herd immunity and increases the opportunity for the non-vaccinated children to develop measles.
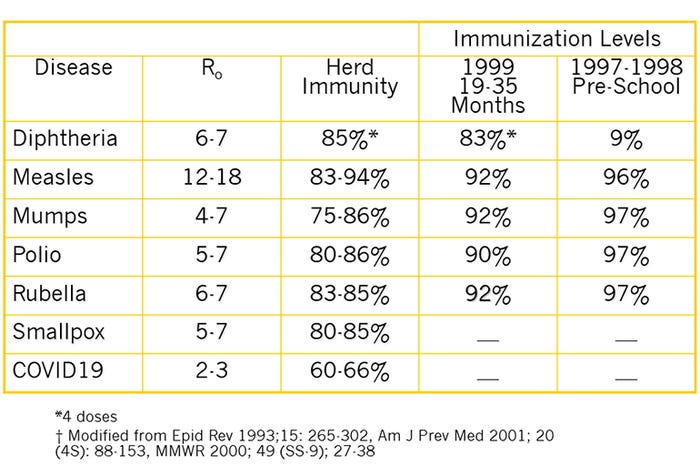
With COVID-19, the level of coverage is considerably lower (Table 1), but without a vaccine, the only opportunity for immunity is from infection, which is why we are socially isolating. Once we have a vaccine, we will be able to develop herd immunity (Figure 1 bottom panel) so individuals not vaccinated (such as newborns and those with chronic illnesses) are offered some protection because the disease has little opportunity to spread within the community.
Summary
Herd immunity is a key concept for both swine and public health. Understanding the epidemiology of spread along with the vaccine coverage in the herd are key components in developing vaccine approaches and protocols. Management particularly of herd density is also key for proper prevention.
References
Rose, N., Andraud, M. (2017). The use of vaccines to control pathogen spread in pig populations. Porcine health management 3(1), 8.
Schulz, K., Staubach, C., Blome, S. (2017). African and classical swine fever: similarities, differences and epidemiological consequences Veterinary Research 48(1), 84.
Source: Chris Chase, who is solely responsible for the information provided, and wholly owns the information. Informa Business Media and all its subsidiaries are not responsible for any of the content contained in this information asset. The opinions of this writer are not necessarily those of Farm Progress/Informa.
You May Also Like



