Methods for assessing bone mineralization
Objective was to evaluate responses to dietary phosphorus, phytase and vitamin D in finishing pigs.
January 12, 2023

In recent years, the occurrence of lameness in growing pigs has increased, leading to an increased incidence of removals and mortality. Lameness is defined as impaired movement or deviation from normal gait. There are many factors that can contribute to lameness, including infectious disease, genetic and conformational deficiencies, impaired physiological development of articular surfaces within joints, and skeletal mineralization. Metabolic bone disease is a common cause of lameness in swine production and can be caused by inappropriate levels of essential vitamins or minerals.
An important component of lameness evaluation is the histopathological examination of tissues including articular surfaces, synovium and growth plates. While these tissues can provide an indication of a variety of pathological processes, such as metabolic bone disease induced by vitamin D deficiency (Madson et al., 2012), they do not always result in a definitive diagnosis. Ancillary diagnostic tests that can be used in a workup of clinical lameness include measures of bone mineralization such as bone ash and serum concentrations of calcium, phosphorus and vitamin D.
Serum 25(OH)D has been the standard for the determination of vitamin D status within swine; however, there has been recent speculation that the activated form, 1,25(OH)2D, may provide additional benefits in assessing vitamin D status (Hurst et al., 2020). There is limited information regarding serum 1,25(OH)2D levels in swine under a variety of feeding conditions.
Bone ash is an established method for measuring bone mineralization. However, questions remain about whether defatted or non-defatted bone ash is the more accurate method. Wensley et al. (2020) compared defatted and non-defatted processing methods to determine their effects on the ability to detect treatment differences. The authors observed that either non-defatted or defatted bone processing methods can be used to determine the bone ash weight and percentage bone ash to assess bone mineralization in nursery pigs although bone ash percentage is greater for the defatted method compared to the non-defatted method.
A non-invasive technique for evaluating mineralization status is the collection and analysis of urine samples. Urinary Ca and P can be measured, and when put in a ratio to creatinine to standardize for potential differences in urine volume, has been shown to be a promising indicator of mineral status. On their own, the presented assays are limited in their ability to diagnose metabolic bone disease and identify the cause. However, when evaluated collectively, the assays can result in a better diagnosis and can lead to intervention. Therefore, the objective of this study was to evaluate the effect of different bone and analytical methods on the assessment of bone mineralization responses to dietary P, phytase and vitamin D in finishing pigs.
Procedures
The Kansas State University Institutional Animal Care and Use Committee approved the protocol used in this experiment. The trial was conducted at a commercial research facility owned and operated by New Fashion Pork (Jackson, Minnesota).
Animal and diets
A total of 882 pigs (PIC TR4 × (Fast LW × PIC L02); initially 73.2 ± 0.7 lb) were used in a 112-day trial. Pens of pigs were fed common diets from weaning until the start of the study. Ca and P levels for all common diets were at or above NRC requirement estimates. Pens of pigs (20 pigs per pen) were then randomized to one of five dietary treatments in a completely randomized design with nine pens per treatment.
The dietary treatments were:
Standardized total tract digestibility P fed at 80% of the NRC requirement estimate (deficient)
STTD P fed at 100% of the requirement (National Research Council requirement) using monocalcium phosphate
STTD P fed at 100% of the NRC requirement with phytase included
STTD P fed at 128% of the NRC requirement (industry level) including 0.14% release from phytase
Diet 4 with additional 2,000 IU/kg of vitamin D equivalency from 25(OH)D3 (HyD)
All diets were manufactured in meal form at the New Fashion Pork feed mill in Estherville, Iowa (Table 1 and 2). For treatments 1 and 2, P levels were met by only using monocalcium phosphate. All other treatment diets had 2,000 FYT/kg of phytase included in the diet to meet the desired STTD P levels, with an assumed STTD P release of 0.14%. Vitamin D was included in all diets via the vitamin premix at a rate of 992 IU/kg. Treatment 5 had an additional 2,000 IU/kg of vitamin D equivalency from 25(OH)D3 HyD, (DSM Nutritional Products, Parsippany, New Jersey). All diets were formulated to an analyzed Ca:analyzed P ratio of 1.20:1. Experimental diets were fed for 112 days.
Pens of pigs were weighed, and feed disappearance was measured every 14 days to determine average daily gain, average daily feed intake and feed to gain ratio. On day 112, one pig per pen (nine pigs per treatment) were euthanized and used for the analysis of bones, blood and urine.
The right and left metacarpal, fibula, 2nd rib and 10th rib were collected from each pig for a total of eight bones per pig. All bones were analyzed using dual-energy X-ray absorptiometry (DEXA) scans, then bone density, breaking strength, bone ash and bone Ca and P were determined. Histologic evaluation of hematoxylin and eosin (H&E) stained sections of the 2nd rib, 10th rib and fibula was performed by three diagnostic pathologists. Hematoxylin stains the cell nuclei a purplish blue and eosin stains the extracellular matrix and cytoplasm pink, with other structures taking on different shades, hues and combinations of these colors. Bones were scored for lesions of failure of endochondral ossification of the physis and microscopic fractures (infractions). Medullary trabeculae and cortical bone thickness was measured.
Ten mL of blood was collected to measure serum chemistry, and 10 mL of urine was collected directly from the bladder to measure Ca, P and creatinine. For de-fatted bone ash all bones were cleaned of tissue and then placed in Soxhlet extractors containing petroleum ether for seven days as a means of removing water and fat. Bones were then dried at 221 degrees Fahrenheit for seven days and then ashed in a muffle furnace at 1,112 degrees Fahrenheit for 24 hours. For non-defatted, the bones were dried at 221 degrees Fahrenheit for seven days and then ashed in a muffle furnace at 1,112 degrees Fahreheit for 24 h. Bone density was measured based on Archimedes principle. After bone ash was completed, the ash was digested and sent to the K-State Research and Extension Soil Testing Laboratory, Manhattan, Kansas, for analysis of Ca and P via ICP.
Growth performance
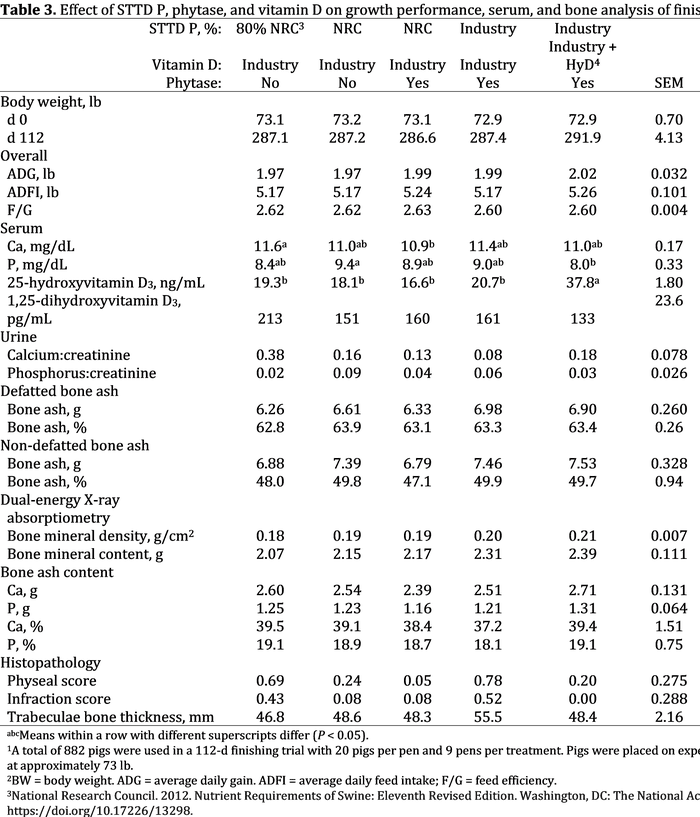
For final body weight and overall growth performance, there were no differences between the dietary treatments (P > 0.10; Table 3).
Serum analysis
For serum Ca, pigs fed 80% of the NRC requirement for P had increased circulating Ca compared to pigs fed NRC P levels with phytase (P P 3 from HyD had increased (P 3 compared to pigs fed industry levels of vitamin D in the diet. 25-hydroxyvitamin D3 is the precursor to the active form of vitamin D and undergoes a bioconversion process within the kidney to form the active metabolite, 1,25-dihydroxyvitmain D3. There was no difference between treatments for circulating 1,25-dihydroxyvitamin D3 (P = 0.147). For urine Ca:creatinine, there was a tendency for a difference between treatments (P = 0.081), but no significant mean separation was observed between treatments (P >0.05). For urine P:creatinine levels, there was no difference between treatments (P = 0.378).
Bone analysis
There were no treatment differences for defatted bone ash percent, percentage non-defatted bone ash, non-defatted bone ash grams, or percentage and grams of bone ash Ca and P (P > 0.10). For defatted bone ash grams, pigs fed industry levels of P had numerically increased bone ash grams compared to pigs fed the other diets, but no significant mean separation was observed (P > 0.05). The fibula had greater percentage defatted bone ash compared to the 2nd and 10th ribs (P P P
The response to treatment for bone density and bone mineral content was dependent upon the bone analyzed (density interaction, P = 0.053; mineral interaction, P = 0.078; Table 5). There were no treatment differences for bone density and bone mineral content for metacarpals, fibulas and 2nd rib (P > 0.05). For 10th rib bone density, pigs fed industry levels of P and vitamin D had increased (P 3 (from HyD) had increased bone mineral content compared to pigs fed deficient P and NRC levels of P with phytase, with pigs fed industry P and vitamin D, and NRC P with monocalcium intermediate.
For histopathological analysis of bones, there were no treatment differences for infraction score, trabeculae bone thickness and cortical bone thickness (P > 0.10). For physeal score, there was a significant difference between treatments (P = 0.040), but no significant mean separation was observed. Numerically, pigs fed 80% of the NRC P requirement and those fed industry levels of P with industry levels of vitamin D had an increased physeal score, representing more lesions of endochondral ossification.
In summary, bone density and bone mineral content responses varied depending on the bone. The difference between bone ash procedures was more apparent than the differences between treatments. Differences in bone density and mineral content in response to P and vitamin D were most apparent with the 10th ribs.
Appreciation is expressed to the Minnesota Pork Board, Iowa Pork Board and DSM Nutritional Products (Parsippany, NJ) for partial financial support of this project.
For more detail, please find a video of this information from the KSU Swine Day:
References:
Hurst, E. A., N. Z. Homer, and R. J. Mellanby. 2020. Vitamin D metabolism and profiling in veterinary species. Metabolites. 10:371. doi: 10.3390/metabo10090371.
Madson, D. M., S. M. Ensley, P. C. Gauger, K. J. Schwartz, G. W. Stevenson, V. L. Cooper, B. H. Janke, E. R. Burrough, J. P. Goff, and R. L. Horst. Rickets: case series and diagnostic review of hypovitaminosis D in swine. J. Vet. Diagn. Invest. 24(6):1137-1144. doi:10.1177/1040638712461487.
Wensley, M. R., C. M. Vier, J. T. Gebhardt, M. D. Tokach, J. C. Woodworth, R. D. Goodband, and J. M. DeRouchey. 2020. Technical note: assessment of two methods for estimating bone ash in pigs. J. Anim. Sci. 98:1-8. doi: 10.1093/jas/skaa251.
You May Also Like



