Prairie strips incorporate diverse, native plant species that offer greater resiliency and stability due to their adaptions to regional landscape.
September 7, 2021

Antibiotic resistance can be defined as the ability of bacteria to survive and grow in the presence of an antibiotic to which they were once sensitive or susceptible. As a result, medicines previously used to treat bacterial pathogens become ineffective, threatening our ability to treat these infections. The emergence of antibiotic resistance is considered a global threat to public health.
Picture a scenario in which you go to work and your coworker is sick. You try your best to avoid them, but by the end of the day, they’ve gotten you sick as well. You go to the doctor, find out it’s a bacterial infection, receive some antibiotics, take them, and get better. Now imagine you’re sick again with the same infection. You go to the doctor, and you receive antibiotics, only this this time you do not get better. Your doctor discovers this is because the bacteria making you sick are resistant to the previously effective antibiotic. Because of this, the antibiotic has no effect on your illness.
Finally, picture a production animal housed in an area concentrated with other production animals “interacting with their coworkers” on a much more frequent and amplified scale. The likelihood that one sick individual could transfer their sickness is greatly increased. To combat this, antibiotics may be administered on a much more frequent basis and to the entire herd rather than just the specific sick individual. Moreover, these animals can outweigh, or even outnumber, humans, meaning the antibiotic dosage required to treat them is often much higher. While the use of antibiotics is a necessary facet of animal production, it also represents a significant evolutionary driver contributing to antibiotic resistance.
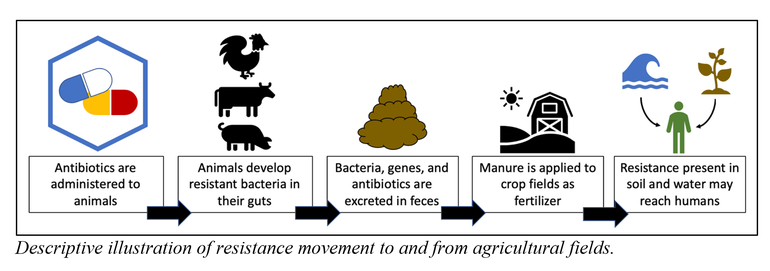
In agriculture, a pathway exists for antibiotic resistance to develop and then spread to the environment. Although antibiotics are our treatment against pathogenic bacteria, they also represent a selective pressure that pushes the development of resistance. Since antibiotics are commonly and necessarily administered to production animals, an opportunity exists for antibiotic resistance to develop in the bacteria present in the digestive system of these animals. As a result, bacteria containing antibiotic resistance, as well as any undigested antibiotics, can be excreted in manure. A common practice is to utilize this manure as a fertilizer and apply it to crop fields. Once applied, recipient soils and downstream waters represent reservoirs of resistance where human exposure and a further exchange of genetic resistance among bacteria can occur.
Although it is not the only source contributing to the spread of antibiotic resistance, it is important that we look at how we can reduce the contribution that agriculture has to this issue. Likely the most impactful way to reduce the spread of antibiotic resistance starts upstream, with the responsible and monitored use of the antibiotics distributed to production animals. This can help ensure that animals aren’t unnecessarily creating an environment for antibiotic resistance development. In addition, conscientious application of manure can help to limit the introduction of antibiotic resistant bacteria, antibiotic resistance genes, and antibiotics into the environment.
Once introduced into the environment, various solutions are considered to limit the transport and persistence of these antibiotic resistant contaminants. One such strategy has come through a partnership with the STRIPS (Science-based Trials of Rowcrops Integrated with Prairie Strips) team at Iowa State University. Currently, research is being conducted to investigate if the use of prairie strips within, and at the edge of, manured fields can help combat the transport of antibiotic resistant contaminants downstream.
These prairie strips are a conservation practice utilizing native perennial vegetation, and they have been researched extensively for their other benefits for over a decade. Unlike many typical vegetative strips, which utilize monocultures of nonnative grasses, prairie strips incorporate diverse, native plant species that offer greater resiliency and stability due to their adaptions to the regional landscape. Presently, research has demonstrated that prairie strips are a cost-effective implementation that can provide agroecosystem benefits including the reduction of water runoff, soil loss, and nutrient runoff, while increasing plant, insect, and wildlife biodiversity. These already identified benefits have promoted the proposal that prairie strips’ uses may extend to antimicrobial mitigation.
Current research goals include:
Evaluating the ability of prairie strips to prevent the movement of resistant bacteria, resistance genes, and antibiotics downstream of manured crop land,
Creating a model that can evaluate the performance of prairie strips and inform field designs, and
Characterizing differences between prairie strip soil and crop soil microbial communities while examining their responses to manure application.
Exciting key takeaways from ongoing experiments include:
The incorporation of prairie strips does appear to reduce the presence of antibiotic resistant contaminants within runoff water,
Antibiotic resistant contaminants from manure don’t dissipate any more slowly within impacted strip soils than within impacted crop soils, and
Preliminary modeling shows that even in very bad case scenarios, in which there is a high concentration of incoming antibiotic resistant contaminants moving with runoff water, current design recommendations for prairie strip widths could provide sufficient reduction to meet official water quality standards
Sources: Michelle Soupir, Iowa State University, who are solely responsible for the information provided, and wholly own the information. Informa Business Media and all its subsidiaries are not responsible for any of the content contained in this information asset.
You May Also Like



