Salmonella: How can we fight ‘smart’ bacteria?
Vaccine efficacy, for instance, is still variable due to the great diversity of Salmonella types that colonize pigs, and it may not completely protect the herd.
August 13, 2019

By Mariana Meneguzzi and Matheus Costa, University of Minnesota Veterinary Population Medicine
If you are a pig producer, you might want to familiarize yourself with the bacterium Salmonella and its importance for the swine industry. After all, it is good to know who your foes are.
In grower-finisher pigs, Salmonella can cause generalized infection (septicemia) or diarrhea (enterocolitis). However, it can also survive in pigs without causing disease. This is a smart way to silently spread around. By colonizing the pig gut without causing any clinical signs, Salmonella can contaminate the environment by being shed in feces. After stressful events, such as transportation to a slaughterhouse or commingling, pigs are more likely to shed Salmonella and consequently contaminate carcasses or infect other pigs.
Currently, Salmonella control in swine herds may be achieved by vaccination or antibiotic therapy. Vaccine efficacy, for instance, is still variable due to the great diversity of Salmonella types that colonize pigs, and it may not completely protect the herd. Antibiotics may also have limited efficacy against Salmonella, due to the bacteria’s intracellular lifestyle (it can dodge both the immune system and antibiotics by living inside pig cells when necessary) and antibiotic-resistance genes.
In addition, farmers worldwide are reducing the use of antibiotics in agriculture. For example, after the rise of colistin-resistance genes that could be transmitted from one bacterium to another, the World Health Organization classified colistin as a critically important antibiotic to human health in 2016. This encouraged the ban of colistin use as a growth promoter and to limit its therapeutic use under veterinary supervision.
Driven by these limitations in treatments, our team has been using an intestinal culture model to evaluate non-antibiotic alternatives to prevent lesions caused by Salmonella in pigs. A segment of intestine from a healthy pig (known as explants) is supplied with specific nutrients, and kept alive in the laboratory (Figure 1-a). This technique allows us to screen potential alternative compounds in a more ethical way, using less animals for research since each segment of intestine can be divided into dozens of explants.
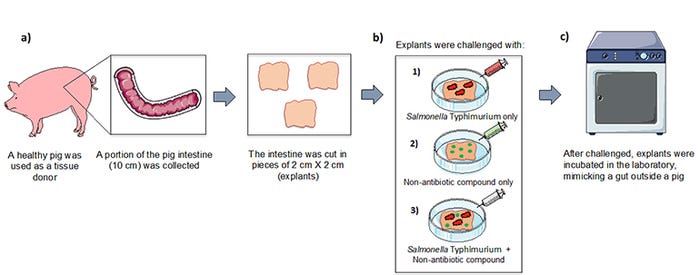
Recently, we conducted an experiment evaluating candidate non-antibiotic Salmonella Typhimurium (which causes diarrhea in pigs, Figure 1-b) treatments. After incubation, we investigated if explants developed microscopic lesions. We measured the ratio of healthy cells, those that kept their original shape even after Salmonella challenge, to unhealthy ones (Figure 2-a and b).
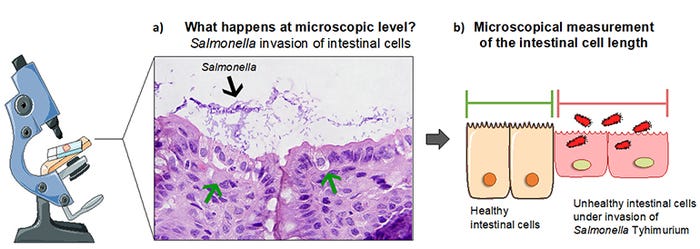
When only Salmonella was inoculated, explants reacted similarly to live pigs (Figure 2-a). However, the treatments evaluated did not seem to affect the severity of lesions. At this point, we are intrigued by this observation. Our current hypothesis is that explants were exposed to an extremely high number of Salmonella (108), which is not similar to natural infection. Infected pigs shed approximately 106 bacteria per gram of feces in the environment, so it is likely that less bacteria are actually ingested by susceptible animals. This overwhelming bacterial load may explain our observations.
Next, we will evaluate the influence of bacterial load, as well as the treatment dose and duration, in protecting the gut from Salmonella. Future findings will be used to reduce the number of animals used in live experiments. In addition, it will accelerate the development of alternative tools for Salmonella control.
Sources: Mariana Meneguzzi and Matheus Costa, who are solely responsible for the information provided, and wholly own the information. Informa Business Media and all its subsidiaries are not responsible for any of the content contained in this information asset.
You May Also Like



