Building herd immunity aids Senecavirus A elimination
Once transmission between pigs on the farm has been stopped, the virus in the environment should be inactivated.
January 8, 2019
By Matthew Sturos and Fabio Vannucci, University of Minnesota College of Veterinary Medicine Department of Veterinary Population Medicine
Discussions about a farm’s disease status will often focus around the detectable presence or absence of a particular infectious agent in or being shed from the animals, usually determined by polymerase chain reaction testing or bacterial cultures. Frequently these results are combined with the presence or absence of antibodies specific for that infectious agent in serum or oral fluids to provide a more complete picture. This approach is logical and useful for many swine diseases because the primary reservoir for these agents are usually the pigs themselves and the agents may have a prolonged period of carriage in infected animals.
However, some agents are also very stable in the environment outside of the host animal, which highlights the importance of also addressing the potential contamination and persistence in the farm environment. One such disease that we as swine farmers and veterinarians have only recently begun to wrestle with and try to understand is Senecavirus A infection.
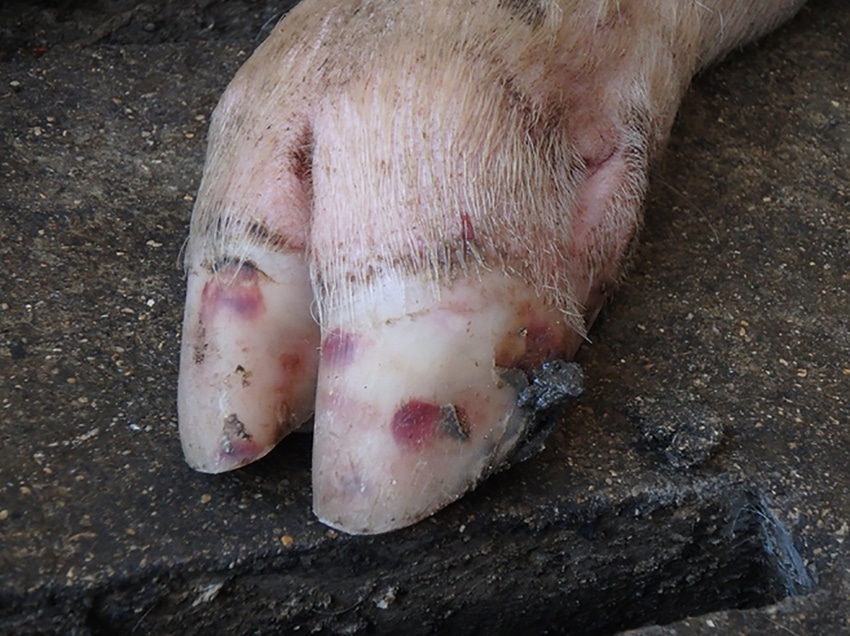
SVA is a virus that can cause blisters, or vesicles, on the skin of the feet, legs and snouts or in the mouths of pigs. One of the current primary concerns about this virus is that the vesicles induced by SVA are indistinguishable from those caused by other vesicular diseases which have important regulatory and trade ramifications, such as foot-and-mouth disease. For this reason, farmers with pigs exhibiting blisters or vesicles, or veterinarians observing these lesions, are required to report these to state or federal animal health officials for further investigation before proceeding with any of the activities discussed below.
While SVA infection doesn’t cause high mortality in adult or growing pigs, these vesicles do have the potential to cause painful feet or mouths and might be initially noticed because of increasing lameness, excess salivation or decreased appetite. The vesicles are delicate and rupture easily, leaving an ulcerated area that will usually heal in one to two weeks. In addition to blisters on the skin, SVA has also been associated with transiently increased pre-weaning mortality on acutely infected sow farms.
There is a large variation reported for the magnitude of these piglet losses between farms. Affected piglets are usually less than 7-10 days old and experience lethargy, anorexia, loss of condition, diarrhea, dehydration or sudden death.
Many of these factors, including increased demand on regulatory personnel, painful feet and skin lesions in affected pigs and increased preweaning mortality, are sufficient for farmers to strive to eliminate SVA from their herds or systems. Elimination at a site level will require a plan to stop viral transmission on the farm, sanitizing the contaminated environment and enforcing strict biosecurity to prevent additional virus introductions in the future. Integral to any elimination plan is appropriate monitoring.
Interrupting transmission of viruses on swine farms is most often accomplished by building strong immunity to the virus in all pigs at the same time, also referred to as “herd immunity.” There are currently no commercial vaccines for SVA in the United States, and so to induce herd immunity farmers need other means. This might be accomplished by exposing non-clinical or naïve pigs to SVA through direct contact with seeder pigs, or pigs known to be infected and shedding, such as those with active or recently ruptured vesicles. Other ways would include utilizing “feedback” of vesicular fluid or infective feces/tissues. Autogenous vaccine products could potentially be used as a means to induce immunity but there is limited information available on this topic. Testing of the quality and quantity of SVA in the feedback material or bodily fluids from seeder pigs by quantitative or semi-quantitative PCR and virus isolation should be performed. Exclusion of other important infectious agents, such as porcine reproductive and respiratory syndrome virus or Salmonella, from these exposure sources is also important.
Once transmission between pigs on the farm has been stopped, the virus in the environment should be inactivated. The virus does not have a lipid envelope surrounding it and thus is not inactivated by detergents or simple washing. Several common disinfectants, including bleach, hydrogen peroxide-based products and quaternary ammonia/aldehyde, have shown high efficacy at inactivating the virus on a variety of surfaces, but the concentrations and contact times needed are variable (Hole et al. 2017; Singh et al. 2017).
After transmission within the farm has stopped and environmental contamination has been addressed, the farm can re-open but attention to essential biosecurity practices will be required to prevent further introductions into the farm. In-depth biosecurity planning is beyond the scope of this column, but important factors to be discussed with the herd veterinarian include the control of entry and movement of animals, people, vehicles, equipment and feed to minimize risk.
Monitoring the elimination plan can be accomplished by intermittent sampling of nasal swabs, oral fluids or feces to be tested by PCR to check for reduction and eventual cessation of active shedding. Many pigs will remain PCR-positive on these sample types for two to four weeks after infection. SVA can be detected in serum by PCR but most pigs are viremic for only a short period of time, so serum samples are most useful to check for SVA-specific antibodies. These antibodies will typically be detectable within one to two weeks after infection and may persist for several months. The environmental contamination load can be assessed by PCR on wipes or swabs collected from surfaces that pigs or employees contact regularly, such as floors, carts, waterers and feeders.
References
Hole, K., F. Ahmadpour, J. Krishnan, C. Stansfield, J. Copps, and C. Nfon. 2017. “Efficacy of Accelerated Hydrogen Peroxide Disinfectant on Foot-and-Mouth Disease Virus, Swine Vesicular Disease Virus and Senecavirus A.” Journal of Applied Microbiology 122 (3): 634–39. doi:10.1111/jam.13361.
Singh, A, S Mor, H Aboubakr, F Vannucci, D Patnayak, and S Goyal. 2017. “Efficacy of Three Disinfectants against Senecavirus A on Five Surfaces and at Two Temperatures.” Journal of Swine Health and Production 25 (2): 64–68.
Source: Matthew Sturos and Fabio Vannucci, University of Minnesota, who are solely responsible for the information provided and is wholly owned by the sources. Informa Business Media and all its subsidiaries are not responsible for any of the content contained in this information asset.
You May Also Like



