The joint meeting will focus on the potential hazards, oversight considerations and labeling of cell cultured food products derived from livestock and poultry.
September 10, 2018
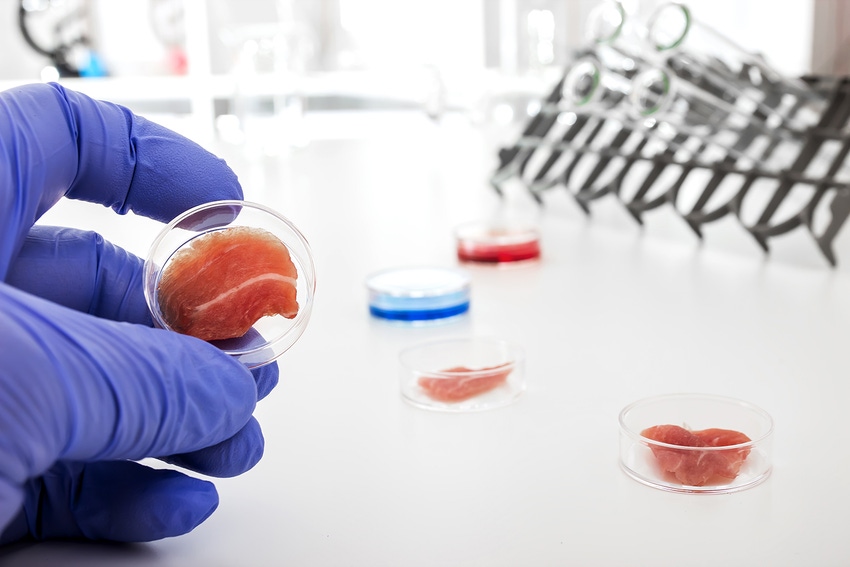
Source: U.S. Food and Drug Administration
The USDA and FDA are teaming up to hold a public meeting on the safety and labeling of cell cultured food products. The joint meeting, slated for Oct. 23-24, will be hosted by the USDA’s Food Safety and Inspection Service and the FDA and will focus on the potential hazards, oversight considerations and labeling of cell cultured food products derived from livestock and poultry.
“This is an important opportunity to hear from the agricultural industry and consumers as we consider the regulatory framework for these new products,” says U.S. Secretary of Agriculture Sonny Perdue. “American farmers and ranchers feed the world, but as technology advances, we must consider how to inspect and regulate to ensure food safety, regardless of the production method.”
“The FDA knows just how vital it is to ensure the safety of our nation’s food supply and the critical role science-based, modern regulatory frameworks are to fostering innovation. Recent advances in animal cell cultured food products present many important and timely technical and regulatory considerations for the FDA and our partners at USDA,” says FDA Commissioner Scott Gottlieb. “We look forward to the opportunity to hold a meeting with our USDA colleagues as part of an open public dialogue regarding these products.”
The first day of the meeting will focus primarily on the potential hazards that need to be controlled for the safe production of animal cell cultured food products and oversight considerations by regulatory agencies. The second day of the meeting will focus on labeling considerations.
Representatives of industry, consumer groups and other stakeholders are invited to participate in the meeting. Attendees are encouraged to pre-register to attend the meeting. Pre-registration is available at the Meetings and Events page on the FSIS website. The meeting will be held on Oct. 23 from 8:30 a.m. to 4 p.m., and Oct. 24 from 8:30 a.m. to 3 p.m. in the Jefferson Auditorium in the USDA building.
Anyone who wishes to submit written comments prior to the public meeting or after the meeting may do so by submitting comments on regulations.gov by Nov. 26. Comments previously submitted to FDA in regard to the July 12 public meeting will also be considered.
You May Also Like



