DON levels in mixed diets up to two times FDA recommended levels may be a strategy for strategic use of DON-contaminated corn in growing pigs provided pigs have sufficient time to develop tolerance and experience “catch up” growth.
June 7, 2018

By Crystal L. Levesque, Ryan S. Samuel, Tofuko Woyengo and Robert Thaler, South Dakota State University; and Kevin Herrick, POET Inc.
Mycotoxin, specifically deoxynivalenol, contamination of corn and corn byproducts, such as distiller’s dried grains with solubles, is a recurring problem and the extent of contamination is dependent on many factors. DON contamination of DDGS has been particularly problematic for the 2016 corn supply and causes vomiting, inducing feed refusal and reducing growth performance in pigs when present in feed at relatively low levels.
The Food and Drug Administration Mycotoxin Regulatory Guidance advisory level for DON in complete animal feed states no greater than 1 part per million DON to minimize risk of feed refusal and reduced growth. As a result, dietary inclusion of DON-contaminated DDGS is limited, especially for swine because they are particularly sensitive. Chemical pretreatment has been used as a mitigation strategy to reduce the effect of DON-contamination in complete feeds. The objective of the trial was to evaluate the effect of pretreating dietary DON-contaminated DDGS with sodium bisulfite on growing pig performance.
A total of 247 growing pigs (55.3 ± 4.6 pounds) were randomly allotted to 18 pens (13-14 pigs per pen). Each pen contained one three-hole feeder and two free-access nipple drinkers. The pigs were fed one of three experimental diets (six pens per diet) for 21 days. The three diets were corn-soybean meal based with 30% mycotoxin-free DDGS (Control), DON-contaminated DDGS (DONDDGS), or pretreated DON-contaminated DDGS (trtDDGS). Levels of DON in each DDGS source and the mixed diets are listed in Table 1. All diets met or exceeded nutrient requirements for growing pigs based on National Research Council (2012) and provided ad libitum during the experimental period. Pig weight and feed disappearance were measured on days 0, 3, 7, 10, 14 and 21.
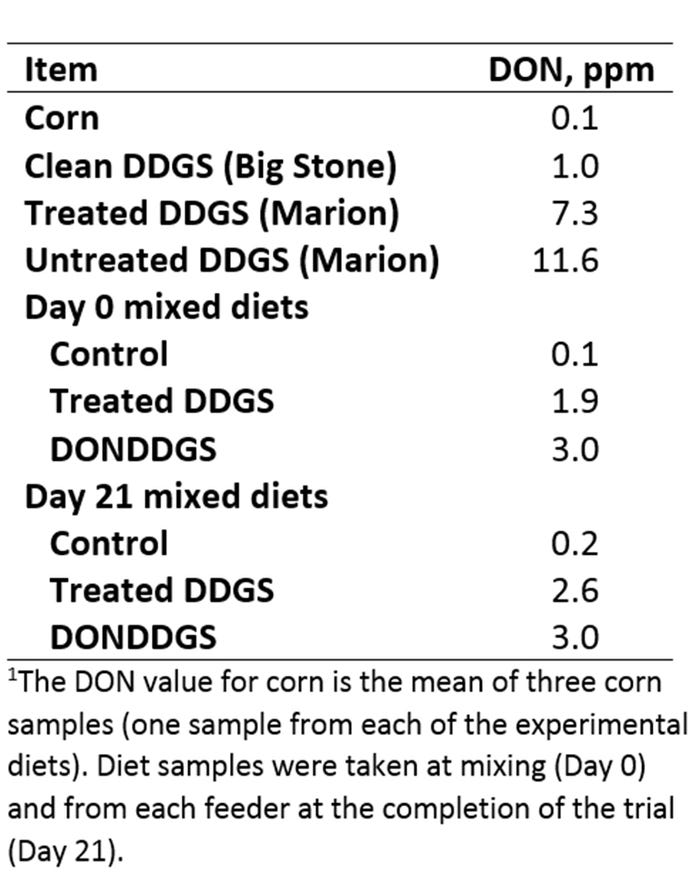
Table 1: Levels of DON in the corn, DDGS sources and mixed diets on Day 0 and Day 21.
Evidence of vomiting was observed within four hours of the experimental diets being available and was observed only in the DONDDGS pens. Vomiting was not observed beyond the first 24 hours of starting the trial; otherwise, pig health was good.
The level of DON in the control diet was below the FDA recommended level. Pre-treatment appeared to be effective at reducing the DON content in DON-DDGS by 37%, resulting in DON content of 1.9 ppm in the treated-DDGS diet. The level of DON appeared stable in the control and DONDDGS diets with some increase in DON levels in the trtDDGS diet by the end of the 21st day.
There was an effect of diet on body weight such that pigs fed DONDDGS diet were 4 pounds lighter than pigs fed Control, and trtDDGS-fed pigs were 1.5 pounds lighter than Control at the end of the 21-day trial (Table 2). Both DONDDGS- and trtDDGS-fed pigs has slower gain in the first three days. There was a tendency for an interaction between weigh period and diet where daily gain was not different between each weigh period in Control pigs and lower in first three days in pigs fed DONDDGS and trtDDGS compared to other weigh periods. Overall daily feed intake for DONDDGS-fed pigs tended to be lower compared to trtDDGS-fed and Control-fed pigs (2.57 versus 2.83 and 2.78 pounds per day ± 0.08, respectively). Overall intake was not different between trtDDGS- and Control-fed pigs.
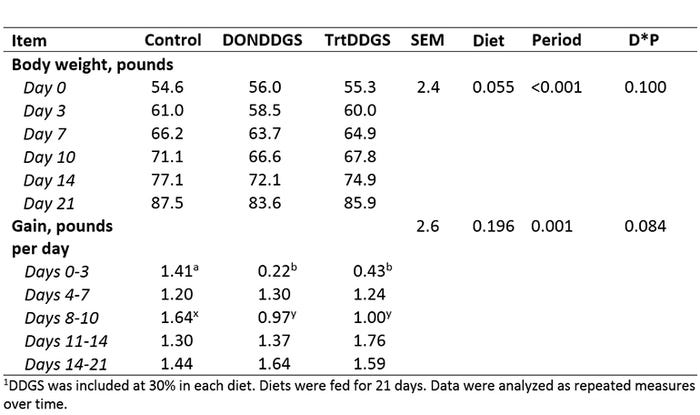
Table 2: Growth performance of growing pigs fed diets containing DDGS with varying levels of DON-contamination1.
There was a tendency for an interaction of diet and weigh period on feed disappearance where feed disappearance increased each week in Control- and DONDDGS-fed pigs and increased from Day 3 to Day 7 then remained constant through the remainder of the trial in trtDDGS-fed pigs (Figure 1). Overall gain:feed was greater (P = 0.049) in Control- versus DONDDGS-fed pigs and tended to be greater (P = 0.062) than trtDDGS-fed pigs. Overall gain:feed was 0.536, 0.409 and 0.416 ± 0.04 in Control-, DONDDGS- and trtDDGS-fed pigs, respectively.
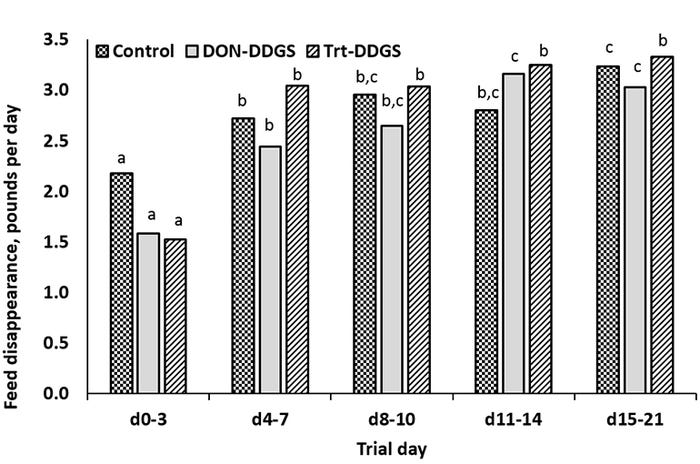
Figure 1: Daily feed disappearance of growing pigs fed diets containing DDGS with varying levels of DON-contamination.
Pretreatment protocols work to convert DON into its less toxic downstream metabolites, but these metabolites may undergo some conversion back to DON in pigs. The slightly higher levels of DON in the trtDDGS diets at the completion of the trial may suggest a similar reconversion of metabolites; however, diets were not analyzed for the presence of DON metabolites which would be necessary to estimate the potential reconversion during the 21-day feed bin storage.
As expected, pigs fed diets with higher levels of DON had reduced daily gains at least in the initial exposure period. Despite the trtDDGS diet containing DON at double the levels recommended by the FDA, pigs fed this diet had reduced gain in the first three days only. Even for the pigs exposed to the highest levels of dietary DON (3.0 ppm, DON-DDGS) some development of tolerance appeared to develop based on similar intake and growth performance in the third study week. However, the improved intake was insufficient to compensate fully for the lower gain in the first week.
Implications
Seasons of high vomitoxin-contaminated corn occur making formulation of “clean” pig diets a challenge. This study suggests that DON levels in mixed diets up to two times FDA recommended levels may be a strategy for strategic use of DON-contaminated corn in growing pigs provided pigs have sufficient time to develop tolerance and experience “catch up” growth.
You May Also Like



