Accurate estimates of amino acid levels that optimize growth and metabolic status can help to improve pork production.
February 9, 2023

Across the U.S. pork industry, about 30 to 35% of pigs born do not reach the market; these mortality and cull losses impose a significant economic and social burden on pork producers. Although there are numerous factors that contribute to these high mortality and removal rates, adequate dietary nutrient intake may help alleviate the problem. However, current methods for estimating nutrient requirements are specifically designed to maximize growth without regard for survival or other performance measures.
The question is whether growth measurements alone are sufficient to develop amino acid recommendations that optimize pig metabolic status, which may aid in overcoming stress and disease and thus improving survivability. To answer this question an exploratory data analysis was performed. The EDA approach is used to conduct preliminary investigations on data to discover patterns, identify anomalies and check assumptions using various data analytics and visualization tools. Following EDA principles, the first step was to check the assumptions on which growth measurements are considered adequate for estimating AA requirements.
Traditionally, it has been considered that animals use amino acids and other nutrients based on their priority of need (Hammond, 1944), with nutrients being used first for functions and tissues essential for the individual's survival (maintenance), then for those required for the production and survival of the animal's offspring (reproduction), animal growth, and finally adipose tissue deposition (Fig. 1A).
Based on this hierarchical model of nutrient use, it has been deemed excessive to supply more AA than is required for maximum animal growth, and thus the linear-plateau mathematical model has been deemed adequate for AA requirement estimation (Fig. 1B). However, there is evidence that AA intake at levels higher than those required for maximum growth (or maximum protein retention) improve various physiological functions such as protein turnover (Young & Marchini, 1990) and transmethylation reactions (Robinson et al., 2016).
Because AA levels above those required for maximum growth improve metabolic status, it was hypothesized that current AA utilization assumptions were incomplete. Consequently, the second step of the EDA was to conduct data mining using data visualization on published data regarding the effects of AA intake at levels above those for maximum growth.
The data-mining approach revealed a consistent pattern that led to the development of the linear-logistic model (Fig. 1D). The linear-logistic model describes the response of growth or protein retention to graded levels of AA intake, and the trend described by this model was observed in pigs of various ages and other species.
The third step in the EDA was to study the physiological relevance of the linear-logistic model. A literature review was conducted for this purpose, and studies whose data illustrated the linear-logistic model alongside other physiological response measures were selected. The literature review revealed that the linear-logistic model captured the dietary AA levels that maximized growth rates, and that animal responses such as milk yield in dairy cows, litter size in pregnant sows, immune response in broiler chickens, and survival in fish were maximized at higher dietary AA levels.
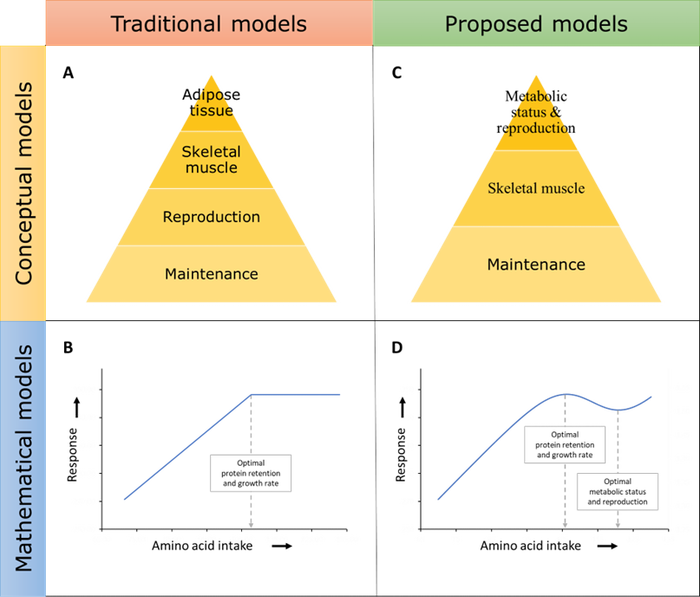
Nevertheless, the consideration that physiological responses related to survival and reproduction are maximized at AA levels above those for maximum growth challenged the concept of the hierarchy of nutrient use proposed by Hammond (1944). The reviewed data pointed to a conceptual model in which animals prioritize lean tissue deposition over metabolic status and reproduction (Fig. 1C).
Wild boar research can help explain why lean tissue deposition may take precedence over metabolic status and reproduction. In a rich nutritional environment, wild boar restrict their movement, whereas in poor environment they increase their mobility in search of food (Morelle et al., 2015). Therefore, as AA supply is reduced, prioritizing lean tissue mass required for mobility and avoiding the increased exposure to predators would provide a short-term evolutionary advantage, but at the expense of metabolic status and, consequently, potential long-term survival.
In conclusion, the EDA suggest that AA requirements estimated using current models result in dietary recommendations that maximize growth and lean tissue deposition but that are insufficient for optimizing metabolic status and thus the potential pig response to stress and disease. The EDA also suggests that growth and protein retention measurements can be used to indirectly estimate AA doses that optimize pig metabolic status and conceivably survival rates when growth and protein retention responses are analyzed using the linear-logistic model.
Finally, accurate estimates of AA levels that optimize growth and metabolic status can help to improve pork production. For example, calculated AA doses for optimal metabolic status can be provided in stages such as the weaning period, where it may be more important to optimize survival rates rather than growth rates, whereas AA levels for maximum growth can be provided in the finishing period.
References
Hammond, J. (1944). Physiological factors affecting birth weight. Proceedings of the Nutrition Society, 2(8).
Morelle, K., Podgórski, T., Prévot, C., Keuling, O., Lehaire, F., & Lejeune, P. (2015). Towards understanding wild boar Sus scrofa movement: a synthetic movement ecology approach. Mammal Review, 45(1), 15-29.
Robinson, J. L., Bartlett, R. K., Harding, S. V., Randell, E. W., Brunton, J. A., & Bertolo, R. F. (2016). Dietary methyl donors affect in vivo methionine partitioning between transmethylation and protein synthesis in the neonatal piglet. Amino acids, 48(12), 2821-2830.
Young, V. R., & Marchini, J. S. (1990). Mechanisms and nutritional significance of metabolic responses to altered intakes of protein and amino acids, with reference to nutritional adaptation in humans. The American Journal of Clinical Nutrition, 51(2), 270-289.
You May Also Like



