Clinical signs remain the same, but flu viruses have dramatically changed
June 19, 2012

Influenza is an important cause of respiratory disease in swine throughout the world. It was first recognized in the midwestern United States in 1918, when the virus moved from humans to swine during the pandemic known as the Spanish flu.
For 80 years, this virus, known as the classical H1N1, remained relatively stable in U.S. swine. Seasonal outbreaks of flu characterized as an abrupt onset of a loud, barking cough, respiratory distress, decreased feed intake and lethargy were caused by the classical H1N1.
During that time, sporadic introductions of influenza viruses from different species or subtypes may have occurred, but they did not become established in the swine population.
Today, swine flu continues to evolve, and the emergence of diverse viruses has generated monitoring and surveillance efforts, which are important factors in maintaining animal and human health.
Influenza Evolution
Although clinical signs of swine flu have remained relatively consistent over the years, the viruses responsible for current outbreaks are much different from the classical H1N1 virus originally isolated from swine in 1930.
Influenza viruses have the unique ability to interchange segments of their genome through a process known as viral reassortment. When this occurs with two viruses of differing subtypes (H1N1 and H3N2, for example), the resulting variant virus may represent an “antigenic shift” that can escape the population immunity from a prior infection, causing outbreaks that can potentially spread locally, regionally and sometimes globally.
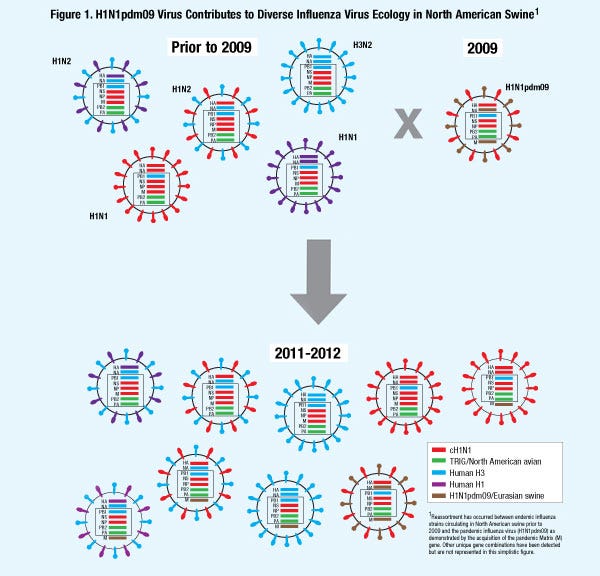
Emergence of a human H3N2 subtype in swine in 1998 significantly impacted the genetic diversity that is recognized in current influenza viruses endemic in U.S. swine. The H3N2 virus was a triple reassortant, meaning it contained a mixture of gene segments derived from swine, human and avian influenza virus lineages.
Triple reassortant influenza viruses that quickly became established in pigs after 1998 are well adapted to the swine host and are consistently isolated from cases of swine flu.
Reassortment continues to significantly contribute to the diverse influenza ecology that is recognized in U.S. swine. After the emergence of the H3N2 virus in 1998, viral reassortment with the classical H1N1 virus resulted in the development of the three subtypes currently endemic in swine — H1N1, H1N2 and H3N2.
Interspecies transmission accompanied by virus reassortment and the emergence of variant influenza viruses in swine have been demonstrated numerous times since 1998. But these occurrences drew little attention from the influenza community.
Novel Pandemic Virus
When a novel pandemic H1N1 virus emerged in 2009 (H1N1pdm09) in the human population in North America, things changed. This virus became infamously known as “swine flu” due to the origin of genes from North American and Eurasian lineages of swine influenza viruses.
Of note, this combination of genes was never recognized in swine before the 2009 human outbreak, but since that time, transmission of the H1N1pdm09 virus from people to pigs has been documented.
The H1N1pdm09 virus is now endemic in U.S. swine and reassortment between contemporary flu viruses and H1N1pdm09 virus is also contributing to the diverse influenza ecology seen in swine (Figure 1).
Approximately 20% of the swine respiratory cases diagnosed at the Iowa State University Veterinary Diagnostic Laboratory (ISU-VDL) are due to influenza A viruses. The vast majority of influenza viruses isolated and sequenced from swine cases at the ISU-VDL are of the H1 subtype.
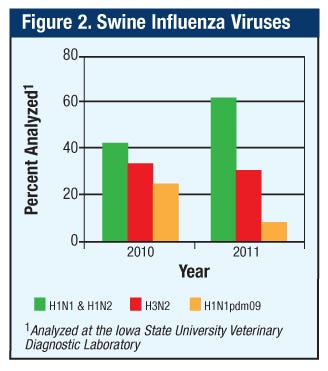
Prior to 2009, no H1N1pdm09 viruses were sequenced at the ISU-VDL. However, in 2010, one year after the pandemic H1N1 outbreak in people, 37% of H1 swine viruses sequenced were H1N1pdm09, confirming the transmission of virus from people to pigs in the Midwest. This value dramatically declined to 11% of H1N1pdm09 sequences in 2011. However, the 2011 percentages do not reflect the number of reassorted viruses that contained genes from the H1N1pdm09 virus (Figure 2).
Reference to the H1N1pdm09 as “swine flu” led to a false alarm over the safety of U.S. pork products and the subsequent loss of revenue due to decreased consumer confidence and decreased exports. The emergence of the H1N1pdm09 virus demonstrated the lack of publicly available influenza surveillance data in human and swine populations prior to that time, especially in some parts of the world.
New Variant Emerges
Surveillance and monitoring of emerging influenza viruses have become increasingly important, from human and swine health aspects. Efforts that help recognize unique features of isolates with pandemic potential may allow time to adequately prepare for preventative measures as well as reduce the adverse economic impact on the U.S. pork industry.
In the fall of 2011, the emergence of a variant H3N2 virus found in humans (H3N2v), a virus with seven genes from the North American triple reassortant swine viruses and one gene from the H1N1pdm09 virus, further emphasizes the importance of influenza surveillance.
USDA Surveillance Program
The U.S. Department of Agriculture (USDA) initiated a surveillance system in 2009 after the emergence of the H1N1pdm09 virus, and recognizing the need for a more integrated and coordinated surveillance strategy in the United States. The system is conducted in cooperation with the National Animal Health Laboratory Network (NAHLN) and will facilitate monitoring of changes in circulating influenza virus strains in swine. The program establishes an objective database for genetic analysis of endemic influenza isolates in swine and assists the development of relevant diagnostic reagents, assays and potential vaccines.
Influenza surveillance in swine is currently conducted through both an anonymous and traceable data entry protocol. Anonymous submissions collected from clinical respiratory cases submitted to participating NAHLN laboratories, like the ISU-VDL, are tested for the presence of influenza. If identified, influenza viruses are isolated and sequenced for subsequent genetic analysis and monitoring of unique isolates. Results are submitted to the USDA anonymously with no owner or submitting veterinarian information. Client-requested diagnostic lab results are reported back to the submitter under current laboratory confidentiality protection.
Veterinarians submitting swine respiratory cases to NAHLN laboratories may voluntarily participate in the traceable surveillance system and receive SIV diagnostic results free of charge. Results are submitted to the USDA with owner and submitting veterinarian information at the permission of producers.
Traceable surveillance provides additional information that aids in detailed analysis of influenza virus genetic diversity and possibly transmission and movement in the swine population. Veterinarians and producers receive free diagnostic test results and sequence information. All virus sequences from the anonymous and traceable cases are deposited in the publically available database, GenBank, anonymously and identified by state of origin only.
Pig Submissions
Samples submitted to the diagnostic lab for influenza testing should be from pigs acutely affected with the disease. Nasal swabs or lung tissues are the currently approved samples for USDA surveillance testing.
Use Dacron swabs for nasal swab submissions and place in a tube of viral transport media prior to shipment. Ship all samples chilled and overnight if possible. The diagnostic lab will conduct tests requested by the veterinarian as part of routine diagnostic processes and report results to the submitter as usual.
Influenza testing requested under the traceable program will be reported to the veterinarian when the virus is isolated and sequenced. Results from the USDA SIV diagnostic workup from samples entered into the anonymous surveillance stream will not be reported back to the submitter and will remain anonymous when reported to the USDA.
Results from submitter-requested tests, conducted at the owner’s expense, are reported back to the client as usual. Contact a NAHLN veterinary diagnostic laboratory with questions regarding this program.
Surveillance Results
The ISU-VDL has received more than 2,000 samples from 22 states for SIV surveillance. Samples submitted from Iowa, Minnesota, Indiana, Illinois, North Carolina and Nebraska account for approximately 86% of samples. All influenza isolates have been subtyped H1N1, H1N2, H3N2 or mixed infections. Among H1 viruses, 13% had H1N1pdm09 HA, NA and M genes (HA-Hemagglutinin, NA-Neuraminidase, and M-Matrix). Remarkably, an additional 49% of other H1 viruses had incorporated the M gene from H1N1pdm09.Among H3 viruses, 46% contained the H1N1pdm09 M gene.
This demonstrates that the H1N1pdm09 virus is co-circulating in U.S. swine, resulting in reassortant viruses with endemic H1N1, H1N2 and H3N2 swine viruses.
In the fall of 2011, an H3N2 variant virus emerged, resulting in 13 human infections in Indiana, Maine, Pennsylvania, Iowa, Utah and West Virginia. Sequencing of these H3N2v from human cases revealed that the M gene was from the H1N1pdm09 virus, but HA and NA genes were from endemic H3N2 swine influenza viruses. This particular combination of influenza genes had not been previously detected in humans.
ISU-VDL surveillance sequence data indicates a virus with HA, NA and M genes closely related to the human H3N2v virus was detected in the swine population as early as Aug. 22, 2011. Similar H3N2 viruses have been detected at the ISU-VDL from Iowa, Indiana, Illinois, Ohio and North Carolina swine since that time.
Prevention and Control
As influenza viruses circulating in swine continue to change, prevention and control efforts have become more complex. Inactivated vaccines used in swine herds may not protect against new variant viruses that emerge through genetic changes. It is difficult for one vaccine to include enough strains to cover all of the multiple subtypes and variants present.
Additionally, it is difficult to update fully licensed commercial vaccines as often as the virus changes in swine.
In recent years, this has spurred increased use of autogenous, inactivated vaccines made from viruses specific to a particular farm. However, these vaccines also may not cross-protect against new strains that unexpectedly infect a herd.
In the future, veterinarians will rely on genetic sequencing more and more to determine if a new strain is present and as a potential predictor of whether a vaccine may cross-protect against a variant virus. The sequence analysis should be paired with serologic tests.
Good animal health management, hygiene and biosecurity remain essential to prevent herds from being infected with new influenza viruses.
Mixing Vessels
Swine can be infected with human- and avian-adapted influenza viruses, resulting in pigs being labeled as “mixing vessels” for generating novel, genetically diverse viruses with epidemic or pandemic potential.
However, humans, other mammals and many species of birds also play a role in the evolution of influenza viruses. This highlights the importance of surveillance in human and avian populations, as well as swine and other non-human mammals.
One can only speculate as to why influenza viruses have changed so rapidly in swine over the past 13 years. However, genetically and antigenically diverse influenza viruses will likely continue to emerge in both the human and swine populations in the future.
Monitoring and surveillance of emerging influenza viruses will require the cooperation of human and animal health officials, researchers and diagnostic laboratories.
Continued surveillance and sharing of information, particularly in the swine sector, demonstrates an ongoing concern, as well as responsibility, that the U.S. pork industry has for its producers and consumers.
Ultimately, the goal is to prevent costly flu-related respiratory disease in swine, protect the pork industry from misleading information, and protect the human population to which the pork industry provides protein.
These early years of the USDA surveillance program will provide the baseline from which the benefits of long-term surveillance for SIV can come to fruition.
Additional authors include Jianqiang Zhang, MD, Veterinary Diagnostic Laboratory, Iowa State University; and Amy Vincent, DVM, National Animal Disease Center.
You May Also Like



