
Every hog producer knows that technology has greatly enhanced the industry’s way of doing business.
One area that is vastly changing in today’s hog industry is biosurveillance. Changes need to be made to be able to react more rapidly in the event of a disease outbreak. That lesson was learned when porcine epidemic diarrhea virus first hit the U.S. swine herd in 2013, and the industry could only react.
Jeff Zimmerman, professor of disease ecology in the Department of Veterinary Diagnostic and Production Animal Medicine, College of Veterinary Medicine, Iowa State University, says producers have a good system in place, but it needs some tweaking.
As a broad definition, Zimmerman says surveillance is data based on testing, but one has to know what they are testing and how. Zimmerman uses Senecavirus A research by Pablo Pineyro as an example. Pineyro had a study of sows that were rRT-PCR-positive for Senecavirus A, even though whether serum, feces or tonsil swabs were tested, 9%, 18% and 27%, respectively, appeared clinically normal (see chart above). Screening of piglets showed similar results: 18% being PCR-positive for Senecavirus A, even though they were clinically normal. Piglets’ screenings of feces and tonsils came back 36% and 27% PCR-positive, respectively. “We need to be in position where we can deal with the subclinical or aclinical,” Zimmerman says.
This leads into Zimmerman’s plea that to make a surveillance work and be effective, it needs to be fast, accurate, affordable and practical. “A long-recognized fact is that we need to have a federal, state and industry partnership,” Zimmerman says.
Though it had been a long-standing feeling that national biosurveillance programs do not work, Zimmerman argues the pieces are already in place; producers just have to know where to start. He quotes George Washington Carver: “Start where you are, with what you have.”
According to Zimmerman, what producers have is “a system with veterinarians in the field; experts in the field where the pigs are. They know what diseases look like, they know how to collect samples and submit samples to the VDLs. Once they’re at the VDLs, we know how to get the results back to them.”
The veterinary diagnostic laboratories are used to heavy lifting. Zimmerman recalls that during the latter days of the pseudorabies eradication program, the ISU VDL was running 1 million to 2 million samples each year. “That’s 5,000 to 10,000 each day; that’s a big testing capability.”
ISU is just one of the many VDLs, and Zimmerman also presented data for the University of Minnesota VDL, which performed 1.4 million tests in 2012. South Dakota State University’s VDL ran just over 400,000 tests in 2013.
“These labs can run a lot of samples, and get the results back out to the field effectively and efficiently through the Web and email,” he says.
In addition to getting the test results back out to the veterinarians and producers in the field, the system is evolving to a shared database between the VDLs. “The power in that concept is so the VDLs will have the data for a coordinated emergency response,” he says. “You could not have built a better system if you had tried to on purpose. … This system has developed on its own, there’s never excessive money. It’s practical and it works.”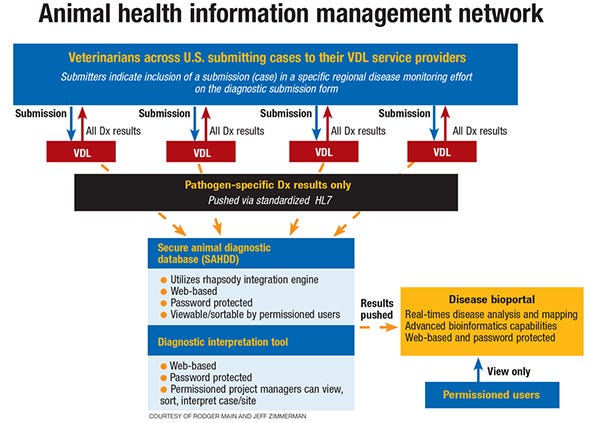
It does work, but it can work even better. “The saying I like is not, ‘the devil is in the details,’ but ‘God is in the details.’ The most beautiful parts of anything are the details, and we have some details to work out here. … We have an unprecedented opportunity to create a supremely effective surveillance system.”
One way to improve the system in place, Zimmerman says, is to collect oral fluids. He says oral fluids “do a lot more work, with a lot less effort than bleeding pigs. … Our vets and our labs know oral fluids.”
Regardless what test the labs decide to run, “we need to be ready to run them tomorrow. We have the capacity [to perform the tests], we have the personnel, but do we have the tests? And right now we don’t,” Zimmerman says. “Especially if you look at foreign animal diseases; we have PCRs in the National Animal Health Laboratory Network labs, but we have limited access to the test that they need to be ready to confront an FAD.”
Zimmerman says chosen tests will need to be available in commercial kits, be able to provide high through-put, and be reproducible and repeatable. “We need to be assured the results will be the same no matter how often the test is repeated,” he says. Lastly, the results of the tests need to be accurate. “The biggest danger is false positives, because you lose the confidence of those you’re trying to help.”
Tests will continually be assessed and improved, and “we need this information now, not after we’ve confirmed a problem. We need to assess our tests, selecting the best tests and encouraging and pushing our diagnosticians to create better tests,” he says.
Once the information is in
Diagnostic data and information are only good if they are in a usable form.
Rodger Main, also with ISU’s VDL, says that in situations where VDLs are testing for diseases that are required by law or rule to be reported to the appropriate state and federal veterinary medical agencies, the VDLs within the NAHLN electronically transfer these reportable disease diagnostic results to the USDA’s Laboratory Messaging Service, which receives and stores the VDL data. This information is then pushed to the Emergency Management Response System, a state and federal authorities’ database system to manage program diseases and program disease events.
This system was used extensively during the 2015 outbreak of highly pathogenic avian influenza. This outbreak led to a four-month sustained response at the laboratory. In Iowa alone, there were 77 HPAI infected premises, with 32 million birds killed, or approximately 40% of the state’s layers.
The core elements of preparedness identified during the PEDV pandemic of 2013-2014 were once again evident during the HPAI experience. Validated diagnostic assays, high-throughput testing platforms, user-friendly and effective sample types for herd-flock level diagnosis, premises identification numbers on VDL submission forms, and electronic messaging of results are important elements necessary to mount a proficient and scalable response to emerging disease outbreaks.
“We proved that we had competent personnel, validated assays, high-throughput testing platforms, user-friendly and effective sample types — both at the farm and at the lab — and we had PINs on diagnostic records, and the ability to message results,” Main says.
Taking a step back and reviewing the effectivity of the process, Main says the quality, completeness, traceability and connectivity of veterinary diagnostic records were identified as areas requiring collective efforts to improve amid the 2015 HPAIV outbreak. Incomplete, inconsistent and/or vague information on the front-end of the diagnostic process (e.g., information included on the diagnostic submission form) was the primary source of confusion concerning the who, what, where, when and why questions associated with veterinary diagnostic records.
“From past experience, we have found that the human-readable formats for premises level identifiers are not really fit for our current way that we handle information in the digital era,” Main says. Premises identification numbers are a unique seven-digit alpha numeric code that serves as the primary premises level identifier and/or point of traceability for official veterinary medical records.
What Main and his team at ISU have figured out is that these PINs can be transformed to a bar code system or incorporated into pre-populated client-specific dropdown menus. Locally they have transformed the front end of the diagnostics system to be more digital. The ISU VDL has implemented a suite of user-friendly methods when submitting specimens to the VDL that allow clients to either use bar codes or a Web-based submission tool. Both options result in the electronic transfer of the full complement of premises or site level identifiers.
At the end of the day, Main says VDLs need to meet the expectations of the stakeholders in the field:
■ competent VDL personnel and infrastructure
■ quality assays and test results
■ turnaround time (same day/next day)
■ electronic distribution of results
■ cost-effective service
■ protect and sustain the continuity their business
These missions are consistent with the local lab, as well as the mission of the NAHLN.
Main says that ISU, in collaboration with the University of California-Davis and Boehringer-Ingelheim, has been working to develop a network capable of “linking VDL submissions, corresponding test results, attending veterinarian insight, and an interpreted health status of farm sites to a highly capable spatiotemporal disease management tool for use in area-regional, veterinary clinic or production system swine health monitoring and control initiatives,” with a desired outcome of providing a user-friendly system for monitoring the health status of swine herds over time. He adds that this information can be used for an individual farm or operation, an entire production system, veterinary practice or area-regional project.
ISU has developed a Web-based platform (Animal Health Management and Evaluation System) that will receive and interpret the swine health data that has been submitted, and then push that information to Disease BioPortal (University of California-Davis) for further analysis and reporting. Disease BioPortal provides permissioned users with real-time disease analysis and mapping.
Main says this network, which is in transition from development to an operational phase, will be piloted as a bureau-based service in 2016, allowing clients to monitor health status and/or diagnostic trends of their pork production operations over time.
Dawn of new age
Disease detection is imperative for any hog producer, and getting a jump on that diagnosis is of utmost importance.
Lindsey Holmstrom, a research assistant professor of epidemiology in the Kansas State University’s College of Veterinary Medicine, feels the Enhanced Passive Surveillance System can bring an important tool for early disease detection into the hands of producers.
Holmstrom explains the main concepts of a syndromic biosurveillance system are to provide automatic or near real-time data collection, analysis and dissemination.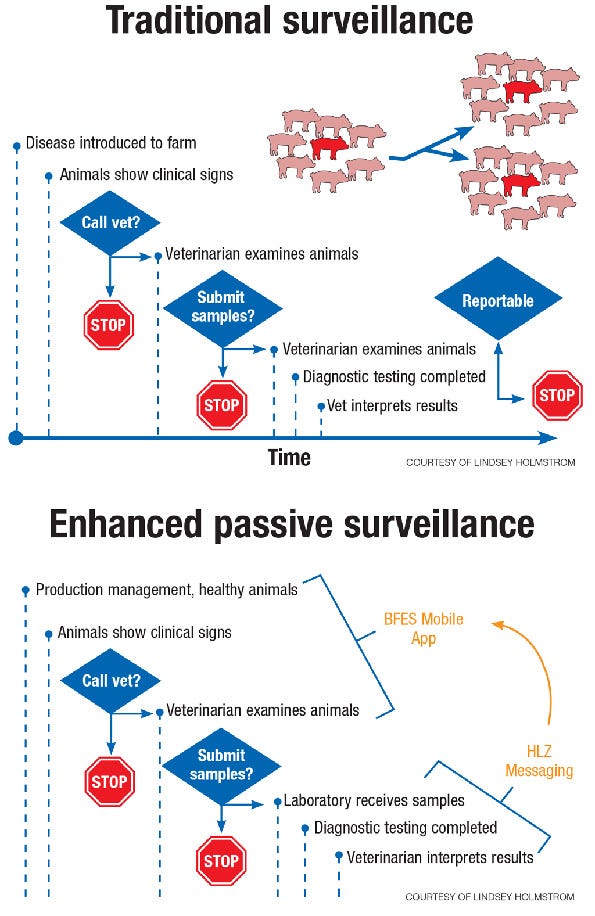
“Technology is trying to bring advances into the hands of producers and veterinarians to help them make better decisions” when it comes to the health and well-being of their swine herd, Holmstrom says.
Though producers and the veterinarians that they enlist to help care for their livestock have become pretty good at detecting health concerns in their herds, a technologically-based surveillance system can present issues before they are physically visible to an individual’s eyes. “The use of data and methods can allow for identification of subtle trends not visible to an individual,” she says. This “heads up” provides indicators to anomaly detection, investigation, quantification, localization, communication and outbreak management.
“Under traditional surveillance, disease may be spreading throughout the farm or an entire production system,” Holmstrom says. “We’re trying to get to an early understanding, and we really need routine collection of disease data, as well as healthy data; what is going on from a clinical view, and to collect that on a real-time basis.”
Common sense says that the earlier a producer or veterinarian can detect a disease anomaly on a farm, the better chance of treatment. Equally important, though, is knowing what is going on around your farm. Holmstrom says by knowing what risks neighboring farms are enduring, producers can then know how to treat their own herds.
With the new emerging or transboundary diseases existing as risks, early detection is even more imperative. “Many have atypical clinical signs and/or clinical signs that mimic endemic diseases,” she says.
Most U.S. producers suffered losses from porcine epidemic diarrhea virus the last few years, so the risk of emerging and transboundary diseases is a very real concern, and that is why Holmstrom sees the ability of this data analysis and monitoring makes this a “really exciting time.”
EPSS was developed by the Institute for Infectious Animal Diseases in 2012, and though in Phase II, Holmstrom says it is still a pilot project.
Phase II will include the expansion to all major livestock and poultry industries, as well as wildlife; develop data confidentiality rules and processes; design industry-customized mobile applications and analyst workstation; incorporate incentives to promote and encourage participation; integrate and evaluate multiple data streams; integrate sophisticated analytical tools capable of processing complex/big data; develop concepts of operations to support the implementation of a national EPS system; perform exercises to evaluate the efficacy/utility of the system and transition a low-cost, low-maintenance system.
EPSS was first made for the iPad, but is now available for most mobile platforms, thus making the information being shared more readily available to producers and veterinarians.
To make any system most beneficial to users, there need to be just that — users.
Holmstrom says there have been efforts made to support participation in the program. Such incentives for participation include access to other developed tools and incentives, as well as a weekly summary report and epidemiological analysis.
An effective surveillance program needs to be an integrated approach linking producers, veterinarians, laboratories, industry, and state and federal officials.
Holmstrom summarizes the benefits of the EPSS:
For veterinarians
■ receive disease trend notifications/alerts from state animal health officials and the USDA
■ share expertise and network on a members-only online discussion forum
■ obtain opportunities for continuing education on the prevention/diagnosis/treatment of diseases affecting their producers/clients
For producers/clients
■ receive subsidized diagnostic testing and Extension support for animal health issues that meet defined clinical criteria
■ support production management and infectious disease control using customizable tools at both the individual farm and integrated company levels
■ obtain increased awareness of disease concerns in the industry that may impact business operations and recommended management practices
For industry
■ detect emerging infectious disease concerns more rapidly than traditional surveillance methods
■ improve communication between key stakeholder groups to manage endemic and emerging infectious disease concerns more effectively
■ protect domestic and international trade markets by identifying geographic areas that are under veterinary observation and free from disease
“Dissemination and decision-making are maybe the most important part of this technology,” she says.
Apps are available for free download on Apple App and Google Play stores, and Web-based platforms are also supported.
About the Author(s)
You May Also Like



