Rectal Swabs a Promising Supplement To E. coli Test
December 22, 2011

Rectal Swabs a Promising Supplement to E. Coli Test
Submitting tissue samples via surface mail may present challenges to swine practitioners due to public health and perception concerns and regulatory restrictions. This study evaluated alternative sampling techniques for Escherichia coli (E. coli) in cases of weaned pig diarrhea.
Fifteen pigs from four sites were sampled. Two rectal swabs, two intestinal swabs and a tissue sample were collected from each pig. Rectal swabs and tissue from each pig were cultured for E. coli, and tissues were evaluated for lesions at the Iowa State University Veterinary Diagnostic Laboratory.
E. coli was isolated from all cases using both rectal swabs and intestinal tissue. Rectal swabs yielded E. coli with both toxin and pili genes in pigs with clinical signs.
However, genotyping results from the rectal swabs and the intestinal tissue did not agree at the pig level in 64% of the cases, suggesting that multiple samples are required to characterize the E. coli population at the farm.
Rectal swabs and tissue samples provide advantages and disadvantages to the practitioner. Tissue samples offer the ability to necropsy the pig and view lesions and other pathogens. E. coli is a pervasive organism in that presence doesn’t always equal disease, so tissue analysis provides an opportunity to verify diagnosis.
Rectal swabs allow healthy pigs to be sampled or acute cases to be pursued where practitioners may be reluctant to euthanize pigs. Rectal swab use helps avoid the need to euthanize the pig thus salvaging its value.
Rectal swabs also allow larger sample size and justify running more antibiotic sensitivity tests due to costs saved from not euthanizing pigs or shipping tissue samples. Rectal swabbing may also provide an opportunity to submit more samples to better characterize the E. coli population without euthanizing additional pigs.
This clinical tool may be used as a supplemental diagnostic method to tissue submission and give a cost-effective means of increasing the sample size in difficult E. coli cases. It will not replace tissue samples from acutely affected pigs as primary diagnostic samples, however.
Researchers: Locke Karriker, DVM and Anna Johnson, Iowa State University. For more information, contact Karriker by phone (515) 294-2283, fax (515) 294-1072 or e-mail: [email protected].
Airborne Spread of Flu Virus is Feasible
University of Minnesota research suggests that swine influenza virus can be detected in aerosols generated from infected pigs and that airborne spread is possible.
Swine influenza virus is an important respiratory pathogen that causes low mortality but high morbidity that impacts pig performance. The virus is known to spread through nose-to-nose contact and droplets, but little is known about the dynamics of airborne transmission within and between herds.
In a study at the University of Minnesota research farm, air samples were collected from 7-week-old pigs experimentally infected and housed in isolation rooms. In addition to the air samples, all pigs were individually sampled daily to correlate individual pig shedding with the likelihood of virus detection in the air.
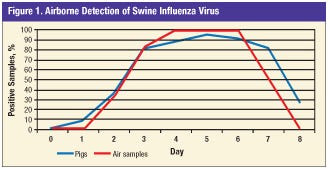
Results indicated that a minimum number of infected pigs need to be actively shedding the virus in order to produce sufficient aerosols to be detectable in the air.
The virus was first detected in the air when at least four of 11 pigs were simultaneously shedding virus. Detection of the virus in air samples under experimental conditions was possible for six days (Figure 1), which coincided with the course of the active infection.
Since the pigs in the study were fully susceptible to influenza infections, pigs with different degrees of maternal immunity against influenza were measured for airborne detection of the virus. Virus was detected in pigs with partial immunity as well as those without immunity, but no influenza virus was detected in pigs that had protective levels of maternal antibodies.
Results indicated that influenza virus may be found in aerosols generated from infected pigs, but that immunity plays a role in the generation of infectious aerosols and therefore in the risk of airborne transmission. More research is needed to understand the role of aerosol transmission under field conditions, according to researcher Cesar Corzo, DVM.
Researchers: Cesar Corzo, DVM, Marie Gramer, DVM, Bob Morrison, DVM, and Montse Torremorell, DVM, University of Minnesota; and Scott Dee, DVM, Pipestone (MN) Veterinary Clinic. For more information, contact Torremorell by phone (612) 625-1233, fax (612) 625-1210 or e-mail: [email protected].
PRRS Vaccine Reduces Shedding in Field Study
One of the crucial challenges in the PRRS regional elimination projects is reinfection of pig units due to area spread of the porcine reproductive and respiratory syndrome (PRRS) virus.
The objective of this study was to evaluate the effect of PRRS modified-live-virus (MLV) vaccine on viral shedding and on dynamics of PRRS infection in pig groups raised under field conditions.
The study consisted of two groups of 1,000 pigs each. Ten percent of the pigs in each room were inoculated with a field strain of the PRRS virus. Rooms featured separate ventilation and strict adoption of scientifically valid biosecurity protocols to avoid movement of pathogens between rooms.
At eight and 36 days post-inoculation (DPI), all pigs in the challenge-vaccine group were inoculated with a PRRS MLV vaccine. Pigs in the challenge-control group were inoculated with a placebo.
Following the challenge, blood and oral fluid samples were collected from each room at 0, 8, 36, 70, 96 and 118 DPI for detection of PRRS virus using polymerase chain reaction (PCR). Screening for PRRS virus antibodies was also performed on blood serum samples using an ELISA (enzyme-linked immunosorbent assay) test.
Tonsil scraping samples were collected from both groups at 70, 96 and 118 DPI. Air samples were collected six times per week from 0 to 118 DPI and tested for PRRS virus using a PCR assay.
Test results suggested that there was no difference in the PRRS infection dynamics as measured by viremia (infection in the bloodstream) and seroconversion between the vaccinated and the control group pigs.
There was no difference in the frequency of tonsil scraping samples testing positive for PRRS by PCR for the two rooms of pigs.
However, the challenge-vaccine group had a significant reduction in the amount of PRRS virus shed in the air and the number of days in which PRRS virus could be detected in air samples. PRRS virus was detected in the challenge-control group for up to 70 days post infection, compared to 45 days in the challenge-vaccine group.
The proportion of oral fluid PCR-positive samples detected at 36 DPI was lower in the challenge-vaccine group.
PRRS MLV vaccines might provide assistance in regional elimination of PRRS virus by decreasing the amount of virus shed from infected pig populations.
Researchers: Scott Dee, DVM, and Joel Nerem, DVM, Pipestone (MN) Veterinary Clinic; Jean Paul Cano, DVM, and Tom Wetzell, DVM, Boehringer Ingelheim Vetmedica, Inc.; Daniel Linhares, DVM, and Montse Torremorell, DVM, Leman Chair, University of Minnesota. For more information, contact Torremorell by phone (612) 625-1233 or e-mail: [email protected].
Young Pigs can be Source of Influenza Virus
Even though swine influenza virus is a common cause of respiratory disease in pigs, information has been lacking on transmission and epidemiology within pig populations.
Anecdotal evidence would suggest that influenza viruses tend to circulate in swine breeding herds for an extended period of time.
New research at the University of Minnesota identified neonatal pigs as a potential reservoir for influenza virus in swine breeding herds. Following intensive sampling in selected commercial herds, neonatal pigs were found to be infected with influenza virus, while the adult animals (sows and gilts) sampled within these populations were not infected.
The prevalence of infection in neonatal pigs was typically low (less than 10%) but tended to be higher as pigs approached weaning age.
Additionally, pigs weaned from an infected breeding herd served as a source of virus for their respective nursery/wean-to-finish herd. This means neonatal and weaned pigs can serve as a source of influenza virus within herds and across herds. This preliminary finding can help guide future research concerning influenza virus epidemiology and control.
Researchers: Matt Allerson, DVM; Montse Torremorell, DVM; and Marie Gramer, DVM, University of Minnesota. For more information, contact Torremorell by phone (612) 625-1233, fax (612) 625-1210 or e-mail: [email protected].
You May Also Like



