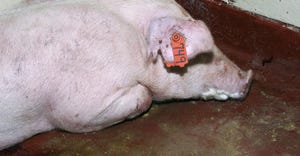
Hog Health
thumbnail
Livestock Management
How to save: Investigate use, frequency of pharmaceuticalsHow to save: Investigate use, frequency of pharmaceuticals
Dive into the data, and ask questions — how many pigs were in the trial?
Subscribe to Our Newsletters
National Hog Farmer is the source for hog production, management and market news











.jpg?width=300&auto=webp&quality=80&disable=upscale)