April 27, 2015

The swine industry faces many challenges when it comes to maintaining the overall health of the herd. At the forefront of these challenges are coronaviruses, a family of RNA viruses with a wide range of infectivity and tropism, spanning both avian and mammalian (including both humans and swine) species. Understanding the similarities and differences about these viruses and their pathogenesis can help prevent viral spread and future outbreaks.
Coronaviruses belong to the family Coronaviridae, which is subdivided into four main genera: alphacoronaviridae, betacoronaviridae, gammacoronaviridae and deltacoronaviridae, which are genetically different and cluster into distant groups in a phylogenetic tree (Figure 1). Five coronaviruses have been detected in swine: transmissible gastroenteritis virus, porcine respiratory coronavirus, porcine epidemic diarrhea virus, porcine deltacoronavirus and porcine hemagglutinating encephalomyelitis virus. Each virus has had a significantly different effect on the industry.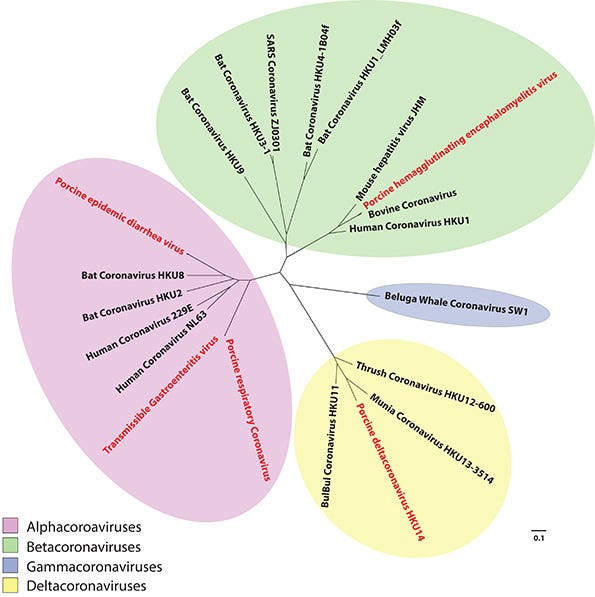 Figure 1: Descriptive CoV Phylogenetic Tree. A phylogenetic tree representation of the evolutionary distances between the different genera of the family Coronaviridae. The four genera (Alphacoronaviridae, Betacoronaviridae, Gammacoronaviridae and Deltacoronaviridae) are denoted by the different colored ellipses. The five porcine viruses are highlighted in red.
Figure 1: Descriptive CoV Phylogenetic Tree. A phylogenetic tree representation of the evolutionary distances between the different genera of the family Coronaviridae. The four genera (Alphacoronaviridae, Betacoronaviridae, Gammacoronaviridae and Deltacoronaviridae) are denoted by the different colored ellipses. The five porcine viruses are highlighted in red.
Whenever multiple viruses from the same viral family affect a host, recombination or the exchange of viral RNA, can occur both within and between a viral species. Recombination within a viral species is beneficial for viral evolution. However, recombination requires co-infection of two different strains in a host, which can be a small subset of CoV infections. Recombination between different viruses is extremely rare. For example, TGEV and PEDV belong to the same genus, and recombination between these two viruses is extremely rare.
Out of these five CoVs identified in swine, PEDV, belonging to the genus alphacoronaviridae, has most recently caused massive losses for U.S. swine. Initially discovered in Europe in 1971, the virus caused epidemics throughout Europe and Asia, has appeared in the Americas, and re-emerged in Europe. PEDV causes severe dehydration, diarrhea and high mortality in piglets. Clinical presentation is less severe in older pigs. Multiple PEDV strains are circulating in different parts of the world, with differing virulence. In Asia, both the classical and pandemic strains of PEDV are present.
In the Americas, two major PEDV strains are circulating – original PEDV and S-INDEL. The original PEDV strain caused many losses to the swine industry and is genetically similar to the pandemic strain while the S-INDEL is characterized by nucleotide insertions and deletions (different types of mutations) in the spike gene, the gene that allows the virus to attach to and infect cells. While original PEDV is the primary target for treatment in the United States, understanding the difference between all strains and how mutations can affect virulence can help lead us to more effective treatment options.
Transmissible gastroenteritis is known as the historical CoV in the United States. Also an alphacoronavirus, the clinical signs of TGEV are identical to PEDV, making laboratory diagnostics essential to distinguish them. Planned virus exposure and infections and biosecurity efforts have helped to limit TGEV infections to mostly finisher pigs. If TGEV enters a naïve sow herd, the virus will cause significant piglet mortality. Despite the fact that PEDV is at the forefront of CoV treatment research, we must remain vigilant as an industry to other CoVs, like TGEV, that could potentially cause similar outbreaks in the future.
Porcine respiratory coronavirus is a variant of TGEV and contains a large deletion in the spike gene, allowing the virus to infect respiratory epithelial cells. There are rarely few, if any clinical signs associated with PRCV infection. A very mild respiratory disease has been described as an infrequent squeal to PRCV infection. Previous exposure to PRCV may help protect against disease caused by TGEV infections. PRCV is considered endemic in swine herds, and pigs are not routinely tested for PRCV.
Porcine deltacoronavirus is another enteric virus and belongs to the deltacoronavirus genus. First reported in Hong Kong, PDCoV first appeared in the United States in Ohio in 2014 and has been detected in 17 states as well as Canada and South Korea. The recent PDCoV strains share an extremely high genetic similarity, suggesting the circulation of a single strain. While PDCoV pathogenesis has been experimentally reproduced, PDCoV infections tend to be less severe compared to PEDV and TGEV infection.
The final virus discussed here is PHEV, belonging to the betacoronavirus genus. PHEV is a ubiquitous virus, and most sow herds have asymptomatic infections. Clinically disease in piglets is associated with gilts failing to get natural pre-farrowing exposure. PHEV infects nervous tissue resulting in two varied clinical presentations. PHEV most commonly causes a central nervous system disease in pre-weaning piglets. With the central nervous system form of PHEV, the virus gains entry into the brain through peripheral nerves and is characterized by lethargy, uncoordinated movements and lateral recumbancy. The gastric form of PHEV occurs most commonly in recently weaned pigs. Clinical signs of the gastric disease include significant vomiting and weight loss due to nerve lesions in the stomach. Morbidity and mortality are typically very low for each clinical form. Pigs over 8-weeks-old are asymptomatic during infections.
Final thoughts
Coronaviruses are a diverse group of viruses causing varied clinical presentations in pigs. PEDV is the current coronavirus of most concern. Knowledge of the existing coronaviruses is essential to differentiate between these viruses for accurate diagnosis. Routine monitoring for viral genetic changes that may affect diagnostic tests and clinical disease presentation are each important for long-term control programs.
You May Also Like



