Investment that the collection and transportation of samples represent underscore the importance of best practices for collection and sample handling to optimize diagnostic timing and accuracy.
September 4, 2018

By Will Lopez, Karen Harmon, Phil Gauger and Daniel Linhares, Iowa State University
Proper sample collection and handling are critical factors for any biological material retrieved from the field for diagnostic purposes. The advent of processing fluids-based sampling to monitor breeding herds for porcine reproductive and respiratory syndrome virus has prompted a rapid increase in this sample type submissions to veterinary diagnostic laboratories in the United States (Trevisan et al. 2018).
Although it is fairly easy and practical to retrieve processing fluids under field conditions from breeding herds, samples require shipment to a laboratory for analysis. The substantial investment that the collection and transportation of these samples represent and the value of the information they provide underscore the importance of best practices for collection and subsequent sample handling to optimize diagnostic timing and accuracy.
The objective of this column is to describe guidelines to collect, store, pack and transport processing fluids samples for diagnostic testing.
Collection
Processing fluids consist of an aggregated sample composed of the serosanguineous drainage derived from the process of piglet castration and tail docking (López et al. 2018) (Figure 1), and should be collected following these steps.
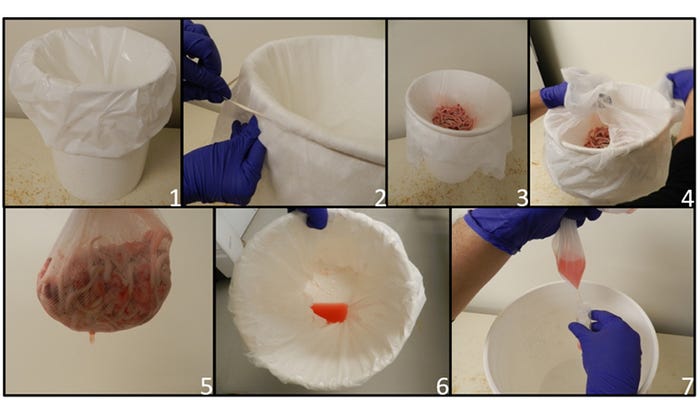
Figure 1: Guidelines to obtain processing fluids from suckling piglets: 1. Plastic bag in clean bucket; 2. Cheese cloth placed and fixed over mouth of bucket to hold tissues and allow fluid to pass through to plastic bag; 3. Collection of tissues from the processing of the litters; 4-5. Tissues are removed after collection; 6. Processing fluid recovered in plastic bag; 7. Fluid transferred from plastic bag into a sterile tube for transportation.
1. Prepare and keep a dedicated bucket for this purpose.
2. Use a disposable plastic bag as lining inside the bucket for fluid collection.
3. Place and affix a piece of disposable cheese cloth (with a rubber band or cord) over the mouth of the bucket to hold tissues and allow fluids to pass through to the plastic bag.
4. Place tissues from tail docking and castration of 3- to 5-day-old piglets during the normal routine of litter processing on the cheesecloth covering the bucket.
5. Remove the cheese cloth with the tissues from the processing of the litters and slightly squeeze it over the bucket to retrieve as much fluid as possible in the plastic bag.
6. Processing fluid will accumulate in the plastic bag.
7. Transfer the fluid into a sterile tube for storage and transportation.
It is very important to use cheese cloth or gauze to strain the tissues and thus obtaining only the processing fluid sample and eliminating the extent of possible pieces of tissue in the sample being sent to the laboratory. Presence of solid material with the processing fluids sample may cause delays for the veterinary diagnostic laboratory to release results due to additional procedures to prepare the samples.
Storing
Duration and temperature conditions of processing fluids storage are critical for the quality of the sample, and diagnostic test accuracy.
Processing fluids can be stored in a regular farm refrigerator (~40 degrees Fahrenheit) or in the freezer (minus 4 degrees Fahrenheit) for up to 15 days with no effect in test performance (López et al. 2018 in preparation), which means that PRRS virus is stable under these conditions. However, multiple freeze-thaw cycles may have a detrimental effect on the ability of polymerase chain reaction-based assays to detect the virus. Thus, it is recommended to avoid freezing-thawing the samples multiple times.
Packing and shipment
Packing the samples is important even when the trip to the laboratory is of relatively short duration. Insulated leak-proof containers and samples lined with a sealed plastic bag are preferred. Also, enough ice or ice packs to keep processing fluids samples chilled until arrival are required to preserve the quality of the sample, the ratio of ice/ice packs to volume of samples should be at least 2:1. Figure 2 shows an example of a well packed set of processing fluids samples.

Figure 2: Packing processing fluid samples for shipment to diagnostic laboratories. The ratio of ice/ice packs to the volume of samples should be at least 2:1.
Conclusion
Regardless of the purpose of the processing fluids submission (i.e. monitoring and surveillance systems, or research), the priority is to preserve and protect the specimens from degradation or decay over time starting at the very first step of sample collection until its use and final storage in the laboratory for further testing if necessary.
Reference
López, Will A., Jose Angulo, Jeffrey J. Zimmerman, and Daniel Linhares. 2018. “PRRS Monitoring in Breeding Herds Using Processing Fluids.” Journal of Swine Health and Production 26(3):146–50.
You May Also Like



