Multiple variants, recombinations and mutations keep the pressure on solving pathogen issue.
December 1, 2020

Porcine circovirus 2 is a DNA virus that can cause severe economic losses in the swine industry worldwide. The introduction of commercial vaccines against PCV2 in the United States and Europe in 2006 was highly effective in controlling PCV2 and improving production.
However, periodical outbreaks were still encountered in vaccinated herds. Investigation of the outbreaks revealed the emergence of a new viral variant, characterized by a unique motif sequence in the capsid gene. The new variant was named PCV2b, while the virus which emerged earlier in the '90s was named PCV2a. The newly-emerged PCV2b variant soon spread rapidly to become the most widely prevalent strain.
A third variant, PCV2d, emerged in 2012 and is now the predominant strain in major pork-producing regions. Besides these PCV2 genotypes, PCV2e has been reported in the United States, Mexico, China and South Korea, PCV2c has been reported in Denmark, Brazil and China, and PCV2f has been reported in China. While PCV2a, b and d have demonstrated similar virulence properties, there is no full characterization of the homologous neutralizing properties among PCV2d strains and the heterologous protection conferred by PCV2a/b vaccines against the new emerging PCV2d strains.
After the emergence of PCV2d, this genotype becomes the most prevalent in the United States. Although this genotype becomes endemic and can be detected subclinically, it is the most common genotype in clinical cases. Data from the Iowa State University Veterinary Diagnostic Laboratory during the period 2016-19 support this description regarding PCV2d prevalence (Figure 1).
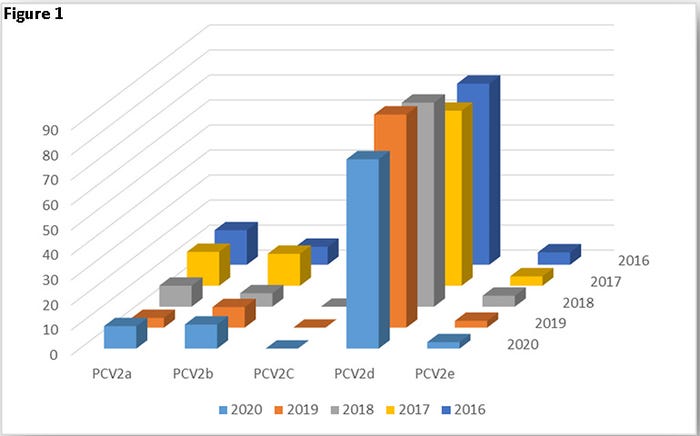
Although at low prevalence, PCV2a and PCV2b cases are still being detected. The main question regarding vaccine efficacy relies on vaccination programs, type of vaccines, timing and doses in addition to piglet versus sow/piglets programs. The endemic nature of all PCV2 genotypes is perhaps the cause of sporadic clinical outbreaks associated with potential vaccine/vaccination failures.
The current commercial vaccines are based on PCV2a/b, by producing a chimeric virus containing PCV1 backbone and PCV2a cap, or baculovirus expressing PCV2-ORF2 gene, or PCV2 subunit vaccines. Over the years, commercial vaccines have proved to reduce the burden of PCV2 clinical disease. Although vaccines can prevent clinical disease, they cannot prevent infection.
Thus, other essential factors need to be evaluated when assessing commercial vaccines' protective role during clinical outbreaks. Assessment of the innate lactogenic immunity might vary depending on the type of vaccine, age and dose used in sows' vaccination program. This strategy might have a direct impact on PCV2-systemic disease (formerly known as postweaning multi-systemic wasting disease) presentation. Vaccination programs that target suckling/weaned pigs might have an impact on protection during the grower-finisher phase. Although antibodies can be detected rapidly after vaccination, true neutralizing antibodies are only developed four to five weeks post-vaccination. Thus, timing, dose or vaccine type in addition to the potential late exposure in endemic farms prompts breaks of PCV2-SD or PCV2-reproductive disease.
The disease
Porcine circovirus-associated diseases can have different clinical manifestations: PCV2 systemic disease that was historically known as postweaning multisystemic wasting syndrome, porcine dermatitis and nephropathy syndrome and PCV2-RD. The evaluation of clinical cases presented at the ISU-VDL with a description of wasting/systemic disease within three weeks postweaning showed a low prevalence from 2016 to 2019.
However, the confirmation of PCV2 on cases of PCV2-SD has increased from 18.5% to 29.6% during the same period (Figure 2). This observation can drive multiple conclusions. However, the most important would be linked to viral evolution and genetic variation. Although most of the clinical cases are associated with PCV2d, phylogenetical analyses showed that this genotype also presents small structural variations that could affect heterologous vaccines' effectiveness. Thus these findings showed that PCVAD-SD is on the rise, and perhaps vaccines and vaccination strategies should be reevaluated.
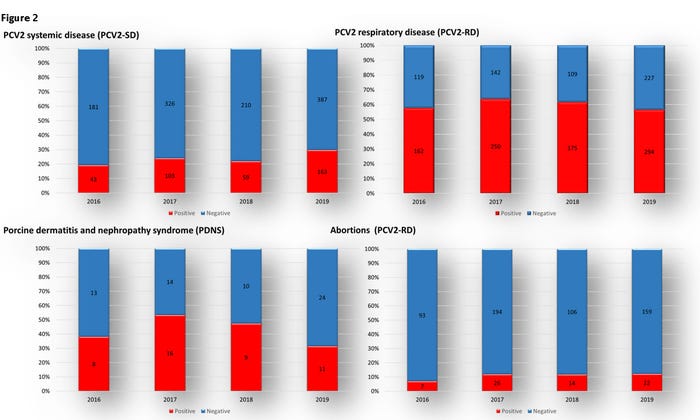
Nonetheless, the implication of PCV2 in the porcine respiratory disease complex, although with small fluctuation over the years, showed that PCV2 was still a main clinical problem in grower-finisher pigs. The summarized data of clinical cases presented for respiratory disease in animals ranging from 8- to 22-weeks of age tested for PCV2 showed that the proportion of PCV2 confirmed cases was about 56% per year from 2016-19 (Figure 2).
Certainly, this data should be evaluated within the context of polymicrobial infections. For example, PCV2 has been present in approximately 30% and 18% of the porcine reproductive and respiratory syndrome virus and influenza A virus cases during the same period. The presence of PCV2 or PRRSV/PCV2 infections are also prone to secondary bacterial infections, with Streptococcus suis and Pasteurella multocida type A being the most prevalent. Although current vaccines and vaccination programs are effective, factors such as duration of lactogenic immunity, time of vaccination or vaccine type should be considered the leading cause of this high prevalence of PCV2 cases associated with PRDC.
Cases with clinical manifestation and lesions of porcine dermatitis and nephropathy syndrome are still being reported. Although the total number of cases seems insignificant, the proportion of PCV2 confirmed cases was 38.1%, 53.3%, 47.4% and 31.4% during the 2016-19 period (Figure 2). Specimens evaluated with this clinical condition are usually in the chronic stage of the disease, making pathogen detection more challenging. Lesions observed grossly and histologically, which most of the time are the trigger of clinical investigations, are likely the result of previous exposure, thus at the time of pathogen detection, viral clearance might already occur.
Therefore, due to this clinical presentation's chronic nature, the real proportion of PCV2 involvement in PDNS should be carefully evaluated, and further investigation is necessary to confirm the real prevalence of PCV2 in cases with PDNS lesions.
Finally, PCV2 is still responsible for cases of reproductive failure. Although typical PCV2 fetal infection lesions are rare, direct detection in fetal tissue obtained during fetal necropsy should be confirmatory. It is essential to highlight that diagnosis of infectious reproductive failures is challenging, and usually, only 20-25% of cases reach a final diagnosis. The proportion of reproductive cases (including abortion and stillbirth) with PCV2 confirmation varied from 7%, 11.8%, 11.6% and 12.1% during the 2016-19 period (Figure 2).
Since 2015, a new member (PCV3) of the porcine circovirus family has been reported as a putative cause of reproductive failure. Although this pathogen's role has not been yet confirmed, retrospective studies showed that the PCV2/3 coinfection rate at the farm level might vary between 30% to 70%.
The diagnosis
Traditionally, the diagnosis of PCV2 has been made based on clinical signs, lesions and viral detection either by polymerase chain reaction or immunohistochemistry. The latter allows associating histological lesions with the presence of the pathogen. This is a critical factor during the diagnostic process of endemic viruses in order to determine causation over pathogen detection. Molecular diagnosis today goes beyond detection and helps veterinarians and producers generate information used to assess clinical disease, make epidemiological decisions, evaluate farm stability, and establish vaccine and biosecurity programs.
Even though the clinical disease is entirely under control due to the effectiveness of commercial vaccines, the number of PCV2 PCR tests requested in the ISU-VDL has increased 89% from 2016 (2,044 tests) to 2019 (3,883 tests). However, the percentage of tests applied to clinical cases is slightly decreasing, varying at 41%, 44%, 24% and 38% of the total PCV2 PCRs performed during the 2016-19 period.
The expansion of sample matrices that can be evaluated by different molecular techniques seems to be the driver of this increment in the number of PCV2 PCRs requested during the last five years. Traditionally lung, lymph nodes (lymphoid tissues) and fetal tissues have been the samples of choice to confirm PCV2 clinical cases. However, nowadays evaluation of oral fluids, colostrum and processing fluids has become a common practice. Molecular techniques applied to these sample types provide different information beyond the clinical diagnosis. Thus, results obtained from these tests need to be interpreted in the right context of the epidemiological situation and the clinical investigation goals.
Numerous molecular techniques at ISU-VDL, including viral typification, quantification, sequencing of specific genes and full-length sequence by next-generation sequencing, are currently available.
Typification by a specific PCV2 genotype PCR has become a common practice. This technique provides rapid information regarding PCV2 genotypes circulating subclinically or responsible for outbreaks of PCVAD. This technique is based on a specific set of primers that independently detect each virus genotype. However, this technique does not provide information on the genetic structure of the genotypes detected. Vaccines, mostly based on PCV2a/b type, can reduce viremia and clinical disease but, most importantly, have been reported to confer cross-protection against other genotypes.
However, the question remains if PCV2a/b vaccines can confer the same protection levels against heterologous strains. Therefore, typing information regarding the strains detected during breaks of PCVAD is an essential tool to evaluate vaccination programs.
Viral quantification has become a useful tool to evaluate subclinical infection and vaccine compliance. The viral load in serum and tissues has provided significant evidence that correlates the severity of lesions, allowing for discrimination between PCV2-causing disease and PCV2-subclinical infection. Due to the ubiquity of PCV2, several studies have proposed that quantitative PCR thresholds in serum varying from 104.7 to 107.43 viral copies per milliliter can be indicative of PCV2-SD diagnosis.
Sequencing of ORF2 is a diagnostic tool that allows evaluation in stable/vaccinated farms of potential new introductions, genetic changes of current circulating genotypes and evaluation of viral dynamics within barn, herd and systems. Viral recombination between different PCV2 genotypes has been reported. Also, PCV2 has shown a high mutation rate. Thus, the ORF2 sequence provides epidemiological/genetic information that evaluates new introductions, mutations or recombination events. A complete historical profile of each system's genomic background is imperative to correlate with new genetic profiles detected in the farm. This information can be directly applied to reevaluate and customize vaccination programs, biosecurity and husbandry protocols.
Highlights
PCV2d is still the most prevalent strain associated with clinical cases
PCV2d has been showing a slow shift pattern in the Cap protein sequence
There has been an increment in the proportion of PCV2-SD confirmed cases
PCV2 is still playing an essential role in cases of pneumonia in grower-finisher pigs (PRDC)
There have not been variations in the proportion of PCV2-RD and PNDS confirmed cases
There is an increase in PCV2 testing request
Importance of monitoring and surveillance to detect potential viral shifts
It is necessary to understand the scope of each molecular diagnostic test to answer the right clinical question
Sources: Pablo Piñeyro, Jianqiang Zhang, Karen Harmon and Phillip Gauger, who are solely responsible for the information provided, and wholly own the information. Informa Business Media and all its subsidiaries are not responsible for any of the content contained in this information asset.
You May Also Like



