Alternative proteins must be produced both sustainably and economically to be widely accepted.
May 6, 2021

For the swine industry to remain agriculturally sustainable in the future, considerable attention must be given to the feed ingredients used in pig diets. Soybean meal is the most common protein source used in livestock diets and makes up almost 70% of the world’s protein meal consumption. Intensive farming of crops can have a significant environmental impact. With swine production expected to continue its growth worldwide, alternative proteins will be needed in order to reduce the environmental footprint. For these alternative proteins to be widely accepted, they must be produced both sustainably and economically. The recent rise in the production of insect meals, algae and duckweed has given swine producers more options for proteins in their rations. Recent work conducted by researchers at North Carolina State University has evaluated a new protein source.
The new product is manufactured by Menon Renewable Products Inc., which is based in Escondido, California. The company has developed a patented process that converts hydrocarbon-based sugars from agriculture-based raw materials and related by-products into a functional animal feed ingredient. Information obtained from the company website states that a variety of unique prebiotics, nucleotides and peptides are created during the manufacturing process which can enhance animal health. The product contains approximately 52% Crude Protein, 5% Crude Fat, 4.35% Crude Fiber, and 8% Ash. It has already been evaluated and used successfully in both the aquaculture and commercial poultry industries, but, until recently, had not been fed to swine.
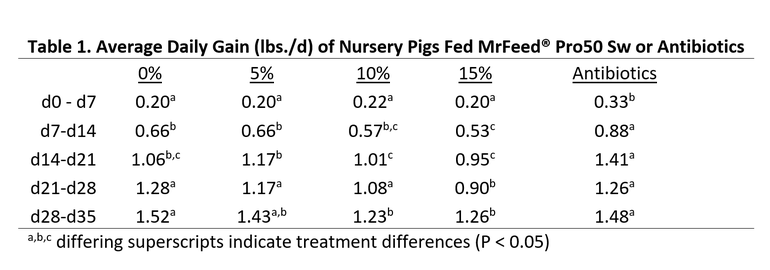
A nursery study was conducted at North Carolina State University with 192 weaned piglets that were offered diets containing either 0, 5, 10 or 15% MrFeed® Pro50 Sw, or a commercial control diet containing antibiotics (carbadox). Experimental diets were fed in three phases. These diets did not contain fishmeal. As MrFeed® Pro50 Sw was added, it replaced soy protein isolate and conventional soybean meal in the diets. Antibiotics were only fed in the commercial diet during the first two phases. All experimental diets were formulated to be iso-caloric, iso-lysinic, and contain the same amino acid ratios. Blood samples were collected at the end of each phase and gut tissue was collected from one pig per pen at the conclusion of the study for histology measurement.
As shown in Table 1 below, pigs fed the commercial diet (with antibiotics) grew faster and were heavier at the end of Phase 1 and 2. However, during phase 3 (when antibiotics were removed from the commercial feed treatment) pigs fed 0 or 5% MrFeed® grew faster than pigs fed the commercial diets. At the conclusion of the 35-day nursery trial, there were no differences in weights of pigs fed antibiotics, 0 or 5% MrFeed®. It is unclear why pigs fed 10 or 15% MrFeed® had reduced growth compared to other treatments. Blood urea nitrogen concentrations of pigs fed MrFeed® treatments were lower than pigs not offered MrFeed® in their diets. This can be an indication of improved protein utilization by the pig. There were no differences in gut histology at the end of the trial. These results demonstrated that when feeding MrFeed® Pro50 Sw at 5% in nursery diets, similar growth performance can be expected compared to using other conventional proteins.
At the conclusion of the nursery study, the pigs were assigned to a finishing trial for approximately 16 weeks, until they reached market weight. Pigs that were fed a MrFeed® containing diet in the nursery trial were maintained on a diet with MrFeed®, included at 2.75%, for the remainder of the trial. Pigs that were not offered a diet with MrFeed® in the nursery were fed finishing diets containing no MrFeed® (control). None of the finishing diets contained antibiotics or other growth promoting ingredients. Growth and feed intake of the pigs was recorded every four weeks.
For the overall finishing trial period, pigs fed MrFeed® exhibited an improvement in feed conversion ratio (FCR) of 4.5% when compared to the control diet (2.06 for control and 1.97 MrFeed®). In addition, pigs offered MrFeed® had a survival rate of 100% during the 16 week trial while those fed the control diet only had a 96% survival rate. While there were improvements in growth performance and mortality of pigs fed the new product, currently there was not an economic benefit to add the product in finishing diets. However, as manufacturing increases and the product becomes more readily available, the economics of its use will likely change.
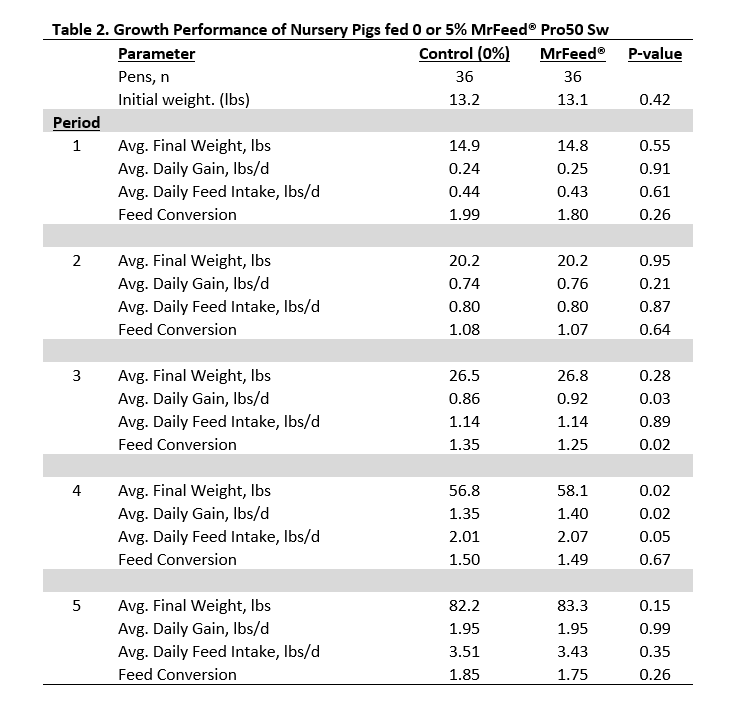
Results of the trials at North Carolina State University were then used to conduct a commercial scale nursery trial in the Midwest. Weaned pigs (n = 1728) were placed into a 2400-head tunnel ventilated, wean-to-finish facility. A total of 72 test pens were used and pigs were placed at 24 pigs/pen for a stocking density of 7.9 ft2 per pig. Two feed treatments were offered (Control or MrFeed® at 5%) with 36 replications per treatment. The treatments were fed in 5 dietary phases over 8 weeks. In Phase 1, MrFeed® replaced all fishmeal in the diet, in Phase 2 MrFeed® replaced other alternative proteins and no diets contained fishmeal. In Phase 3 and 4 MrFeed® only replaced soybean meal and crystalline amino acids as there were no alternative proteins added to these diets. During Phase 5 all pigs were fed a common diet. Pigs were provided feed and water ad libitum throughout the study.
Results indicated that inclusion of MrFeed® Pro50 Sw at 5% in the nursery diet Phases 1 and 2 showed no difference in performance and may be a suitable replacement to other specialty proteins typically included in nursery pig diets. Adding MrFeed® Pro50 Sw at 5% in nursery Phases 3 and 4 provided a statistically significant advantage to growth and FCR, however, it did increase the costs of those diets. There were no carryover effects of feeding MrFeed® in earlier nursery phases as evidence by similar growth reported in Phase 5 when pigs were fed a common diet.
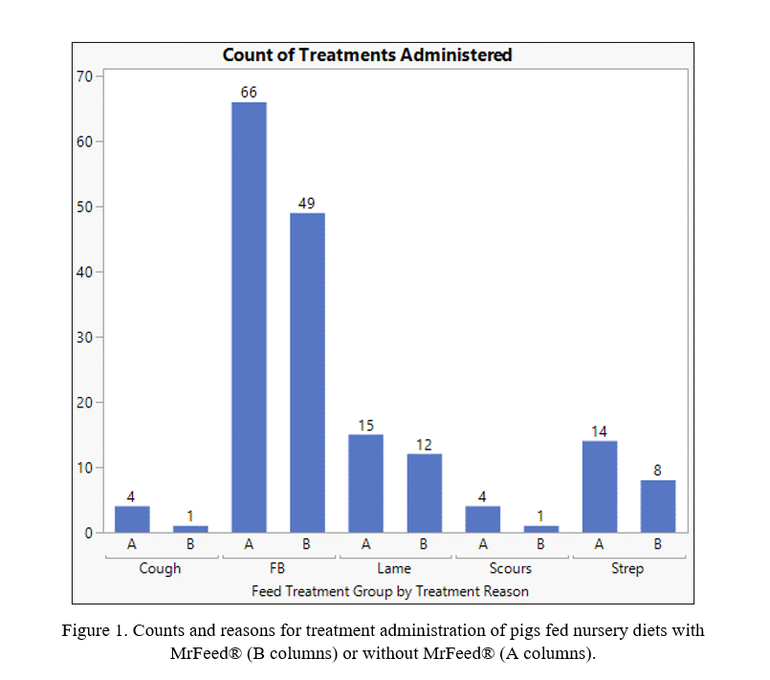
When health of the pigs was observed, it was found that pigs fed MrFeed® Pro50 Sw had reduced number of mortalities (nine total, 1.3% mortality) compared to the control treatments (18 total, 2.7% mortality). Use of MrFeed® also reduced the need for treatment due to ailments such as respiratory illness (strep), diarrhea (scours) and fall back pigs (Figure 1). These improvements in health and livability may help to offset the higher cost of the rations containing MrFeed®.
The commercial trial agreed with research at North Carolina State University, showing that MrFeed® was a suitable alternative protein source for nursery diets. The anecdotal evidence of improved mortality led to further questions about the ability of the product to improve the health of pigs. To answer these, a challenge study was conducted with 50 pigs with a starting weight of approximately 87 lbs. The pigs were placed either four or five pigs per pen and fed a standard commercial diet or the same diet with 5% MrFeed® replacing typical protein sources. Pigs were fed the treatment diets for 21 days prior to challenge with Mycoplasma hyosinoviae on day 0 and PRRS virus on day 7. Pigs were then routinely checked for health and growth performance for 56 days. During the 56 d growth period, pigs fed MrFeed® had zero mortality compared to four mortalities on the Control treatment. Also during this time, pigs fed MrFeed® had greater (P < 0.05) ADG (2.06 vs. 1.75 lbs/d) and ADFI (5.21 vs 4.64 lbs/d) during the challenge period compared to pigs fed the Control diet. Further analysis of blood samples taken from pigs found that white blood cell counts as well as absolute counts of segmented neutrophils and lymphocytes were numerically higher for pigs fed MrFeed®. While blood sample analysis was not statistically different between treatment groups, results could indicate a heightened immune activation for pigs given the alternative protein source. These blood parameters need more replication with a larger group of pigs. This observation may suggest a possible reason why improvements in health and mortality have been recognized in the current and previous trials.
MrFeed® Pro50 Sw has been shown in various trials to be a quality alternative protein source for swine diets. When fed at 5% in nursery rations, it provides similar growth performance to other alternative proteins. Further work is needed to understand how the product may improve health status of pigs. As manufacturing capacity of the product continues to increase, it should become more readily available to swine producers worldwide.
Source: Jonathan Holt, North Carolina State University, who is solely responsible for the information provided, and wholly owns the information. Informa Business Media and all its subsidiaries are not responsible for any of the content contained in this information asset. The opinions of this writer are not necessarily those of Farm Progress/Informa.
You May Also Like



