Research emphasizes nutrient balance and maternal nutrition’s profound impact on bone development in growing pigs.
May 18, 2017

Source: Zinpro Corp.
Recent research investigating bone and cartilage development in pigs is pointing to a link between lesions of mineralization and osteochondrosis, one of the most common causes of lameness and secondary joint disease in pigs.
“Classically, we have thought of bone-related lameness as either a cartilage or a mineralization problem,” says Laura (Rortvedt) Amundson, Ph.D., postdoctoral fellow at the University of Wisconsin-Madison. “Based on our research, there’s potentially more overlap between cartilage development and mineralization than previously thought. Although breakdowns can occur in either process, it is important to think of them as coupled processes instead of separate issues.”
With the development of a vitamin D deficiency-induced kyphosis pig model at the University of Wisconsin Swine Research and Teaching Center, Amundson and her colleagues are gaining new insights into the relationship between vitamin D, calcium and phosphorus. While vitamin D, calcium and phosphorus are highlighted in this research, it is important to note that zinc is another nutrient that’s known to interact with calcium and phosphorus in swine diets. Results from this vitamin D research illustrate the importance of nutrient balance, while also highlighting potential consequences caused by nutrient imbalances. Study findings also reinforce the importance of a sow’s diet during gestation and the profound impact it can have on offspring bone development and growth.
A mixing error triggers new scientific inquiries
A few years ago, the UW SRTC herd experienced an outbreak of kyphosis. Signs of the problem were first seen in growing pigs that had been fed diets containing marginal amounts of calcium and phosphorus (Figure 1). Investigation eventually determined that the outbreak coincided with a period in which a pre-mix deficient of vitamin D3 was fed to the entire herd, including the gestating sows that produced the symptomatic pigs.

Figure 1: Growing pigs from the University of Wisconsin Swine Research and Teaching Center that exemplify kyphosis symptoms.
Since then, researchers performed a series of studies where kyphosis was induced in growing pigs under controlled conditions by manipulating maternal dietary vitamin D supplementation and neonatal pig dietary vitamin D, calcium and phosphorus supplementation. The marginal calcium and phosphorus levels included in the diet to induce kyphosis were not dramatic deficiencies.
According to Amundson, the deficiencies were expected to cause poor bone mineralization and lameness, but not all of the findings could be explained by the classic vitamin D, calcium and phosphorus regulatory pathway. The identification of a vitamin D receptor in bone cells and a newly discovered bone cell-produced hormone, fibroblast growth factor 23 (FGF23), offered new insights into the roles of vitamin D and phosphorus in bone development. Amundson and her colleagues at UW-Madison were the first to report FGF23 expression in pig bone.
Study design essentials
Amundson and Tom Crenshaw, Ph.D., professor of animal science at UW-Madison, designed a follow-up study to evaluate how maternal dietary vitamin D concentrations affect the subsequent growth and bone traits in young pigs. First-parity gestating sows were randomly assigned to one of three dietary treatments (fed from breeding through lactation) that varied in supplemental vitamin D: 0 (–D), 325 (+D) or 1,750 (++D) IU of vitamin D3 per kilogram of diet.
Pigs were weaned at three weeks and randomly assigned within a litter to one of four nursery treatment groups. After consuming an adjustment diet containing no supplemental vitamin D for one week, nursery pigs were fed diets containing either 0 (–D) or 280 (+D) IU of vitamin D3 per kilogram of diet and either 95% or 120% of the phosphorus requirement for 10- to 20-kilogram pigs. No phytase was added to any of the treatment diets. In addition, the diets provided 100% of the growing pigs’ calcium requirement. The treatment diets were fed for four weeks.
Whole body and femur dual-energy X-ray absorptiometry scans were obtained at zero, three and eight weeks to determine bone mineral content and bone mineral density. Researchers also assessed bone mechanical properties and gene expression (note: only results related to FGF23 are included).
Expected and unexpected findings
Differences in pig body weight at week zero were not detected, but were noted at three weeks and in zero- to three-week weight gain (P < 0.05). At three weeks, pigs from –D sows had a 10% decrease in weaning weight and an 11% decrease in weight gain compared to pigs from +D and ++D sows.
Over the 35-day nursery period (from three to eight weeks), differences due to nursery diet treatment were found in body weight gain. Pooled across maternal dietary treatments, pigs fed nursery diets containing no supplemental vitamin D (–D) had an 11% decrease in weight gain (P < 0.05). This finding was not expected, given the short time frame during which the nursery diets were fed. Pigs fed the 120% diets had increased weight gain when fed marginal vitamin D diets (+D), but decreased weight gain when fed –D diets (P < 0.05), suggesting that the slight increase in dietary phosphorus further compromised pig growth.
Differences due to sow diets were detected in pig whole-body BMC and BMD at the end of the nursery period (P < 0.05; BMD results shown in Figure 2). Pooled across nursery treatments, pigs from –D sows had a 19% decrease in BMC and a 25% decrease in BMD compared with pigs produced by +D and ++D sows. In addition, pigs fed –D 120% diets had reduced BMC and BMD if produced by –D or +D sows, but had increased BMC and BMD if produced by ++D sows (P < 0.05). This suggests that feeding excess dietary phosphorus to pigs produced by sows that received no (–D) or marginal (+D) supplemental vitamin D did not compensate for the lack of vitamin D in the nursery diet.
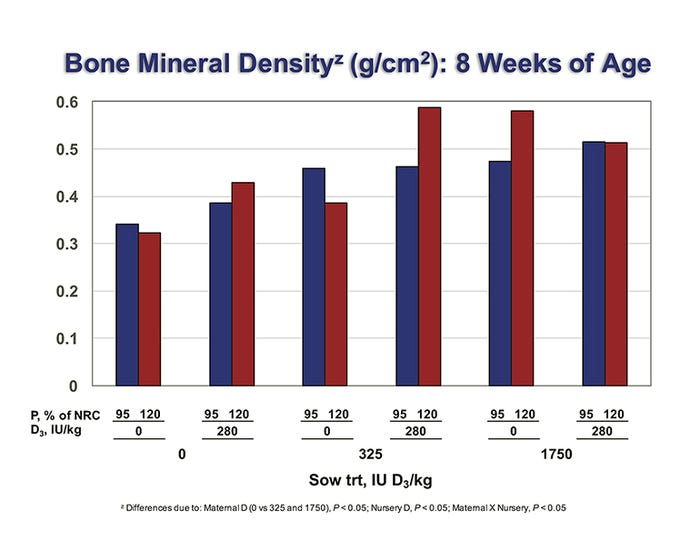
Figure 2: Effect of different vitamin D and phosphorus levels in sow diets on the bone mineral content and density of their piglets.
Bone as an endocrine organ?
A recently discovered peptide hormone, FGF23 is produced by bone cells and is responsible for phosphorus homeostasis. Active vitamin D or phosphorus increases FGF23 production in bone, which then acts on the kidneys to decrease phosphorus reabsorption (i.e., increases urinary phosphorus excretion) and, subsequently, circulating phosphorus. FGF23 also acts to reduce vitamin D activation.
“FGF23 provided a direct link between phosphorus and vitamin D,” Amundson explains. “There’s a negative feedback relationship between FGF23, vitamin D and phosphorus, with the role of FGF23 being to keep phosphorus and vitamin D at homeostatic levels.”
In this study, bone expression of FGF23 was upregulated only in pigs at eight weeks if the pigs were fed +D or 120% nursery diets (P < 0.05). These findings are consistent with the endocrine role of bone in FGF23 production.
Implications for real-world pork production
“Excess nutrients can contribute to bone-related problems as a result of nutrient interactions,” Amundson says. “Whether we’re talking about calcium, phosphorus, vitamin D or another essential nutrient such as zinc, more is not always better. We must think about the interactions among all of the nutrients in the diet.”
According to Amundson, the difference between nutrient deficiency, adequacy and excess supplementation can be very small. Research results involving vitamin D, calcium and phosphorus illustrate that a disturbance in either direction may have severe consequences.
“The insult of a deficiency — or even an excess — in the sow diet can potentially program the resulting pigs to respond differently to dietary inputs later in life,” Amundson states. “We need to understand the offspring’s background and how that might affect their response to different dietary or disease challenges. So it’s important for producers to know what’s going on in their herds’ diets and to pay close attention to sow diets — because of the impact diet can have on offspring.”
When formulating swine diets, producers and their nutritionists should remember that both the amount and the form of a nutrient are important. For example, research has shown the form of zinc contained in AvailaSow and AvailaZn from Zinpro (zinc amino acid complex) is unaffected by common antagonists in the diet. This means more of the zinc is delivered to the animal – which supports improved animal wellness and performance.
For more information, contact a Zinpro representative.
You May Also Like



