A fecal qPCR test may be the future of testing infection levels of Lawsonia intracellularis in pigs at the farm level.
July 24, 2017

Sponsored Content
Nicknamed the “hidden profit robber,” you can’t always see it, but subclinical ileitis is likely causing issues in your pigs. But, what if you could detect those subclinical infections and treat them before they become an issue?
Pigs infected with Lawsonia intracellularis, the bacteria that causes ileitis, will shed the bacteria at varying rates, depending on the level of infection. Past research has shown that shedding rates correlate to the severity of lesions found in the intestinal tract, which then relates to the levels of performance loss.
Monitoring the levels of L. intracellularis shed in the herd can be an important tool in understanding the infection status of the herd. In 2015, the Boehringer Ingelheim Health Management Center Diagnostic Laboratory in Ames, Iowa, developed the first commercially available quantitative polymerase chain reaction test in the United States, which measures L. intracellularis in the feces. The company then studied the capability of this test to also monitor subclinical levels of ileitis.
“This qPCR test is so sensitive it can detect down to one log of L. intracellularis in the feces,” says Jessica Seate, DVM, technical manager for enteric vaccines. “Our goal for this study was to determine if we can use qPCR testing to develop a matrix to estimate the performance loss in that herd, depending on the level of shedding the test detects.”1
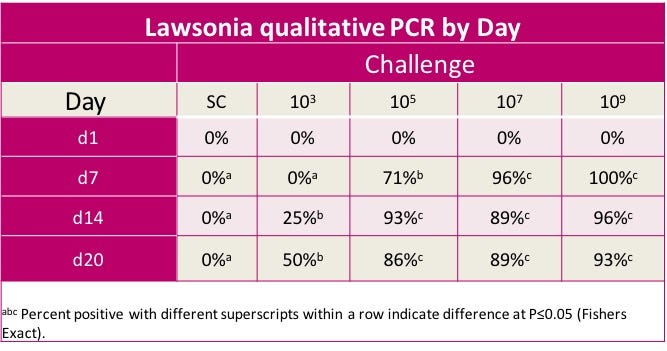
To execute the study, pigs were divided into five groups of 28 pigs each. Groups 1-4 were challenged with increasing levels, or logs, of L. intracellularis, while Group 5 was kept as the control group. Pigs were weighed and challenged on Day 0, and weighed and necropsied on Day 21. All pigs were non-medicated and non-vaccinated. Fecal samples were collected on Days 1, 7, 14 and 20.
“We ran this study for three weeks, because based off literature, we know that is when we should see a peak in lesions in the intestines,” Seate says. “We also took fecal samples every week throughout the study. And, what we found is that groups receiving the higher concentrations of L. intracellularis were almost all shedding the bacteria by Day 7. However, in groups receiving the lower challenge doses, we also saw shedding between Days 14 and 20.”
Study results and conclusions
The pigs were weighed daily throughout the study and results in the graph below showed that the daily and total weight gains in pigs is inversely related to the level of L. intracellularis challenge. The reductions in the average daily gain also correlated to the subsequent examination of lesions in the intestinal tract.
“If you look at the results of the ADG you can see that it declines as the challenge dose increases. There were significant differences in ADG between the higher challenge groups and the control group,” Seate adds.
Performance – Weight Gain d1 – d21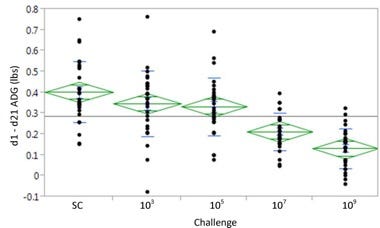
While the groups receiving the higher challenge doses did see more severe reactions, pigs in the lower groups also faced some disease pressure. To measure the effects of ileitis, the pigs were all necropsied on Day 21 to study the severity of lesions in the intestines.
“Research has shown a strong correlation between increased pig shedding of L. intracellularis bacteria and the severity of intestinal lesions,” Seate says. “We also found that to be true when we measured macroscopic and microscopic lesions in the ileum, cecum and colon. We were able to show a positive correlation to the increasing dose level in the L. intracellularis challenge.”
Seate says the next steps will be to conduct validation studies on the L. intracellularis qPCR tests in the field to determine the L. intracellularis status on farms. “With this information, we’ll be able to begin to develop a matrix using the qPCR test to determine if subclinical or clinical ileitis is present in the swine herd. And, this test can be completed without euthanizing piglets or sows.”
Sources:
1Seate, J, et al.; A fecal quantitative polymerase chain reaction (qPCR) assay for Lawsonia intracellularis and the correlation to pig performance in Lawsonia intracellularis challenge pigs. ESPHM 2017, Prague
©2017 Boehringer Ingelheim Vetmedica Inc.
About the Author(s)
You May Also Like



