Study demonstrated the addition of 10% highly fermentable soluble fiber mitigated experimentally induced colibacillosis whereas feeding highly insoluble fiber with low fermentability aggravated colibacillosis
July 27, 2017

By Qingyun Li, Nick Gabler and John Patience, Iowa State University Department of Animal Science; and Eric Burrough, Iowa State University Department of Veterinary Diagnostics and Production Animal Medicine
Pigs are subject to numerous health threats as they undergo various stresses at the time of weaning. At the same time, their immune system is not fully developed. Diarrhea is one of the common problems in a newly weaned group, causing substantial economic losses for producers due to the increased mortality and morbidity. Certainly, many factors can lead to post-weaning diarrhea, such as poor environmental hygiene, inappropriate feeding and infectious pathogens. Piglet diarrhea caused by Escherichia coli is one of the most common cases, also called colibacillosis.
Post-weaning colibacillosis usually occurs between four and 14 days post-weaning. It is caused by enterotoxigenic E. coli (ETEC), mainly F4 and F18 strains in piglets. ETEC adhere to the small intestinal epithelium via fimbriae, and subsequently secrete enterotoxins into enterocytes that eventually result in diarrhea. Infected pigs can die from extreme dehydration due to the hypersecretion of fluids including water.
Historically, antibiotics have been routinely used to prevent or control colibacillosis. However, with the growing concern over antimicrobial resistance and new Veterinary Feed Directive regulations, interest in seeking effective alternatives, such as pre-/pro-biotics and dietary fiber, has increased significantly.
Dietary fiber is indigestible carbohydrates that cannot be broken down by enzymes naturally produced by the pig; however, they can serve as a source of energy for intestinal microbes. In this way, fiber can regulate the gut microbial population. A few studies have been conducted in Europe to evaluate the impact of dietary fiber (e.g. pearl barley and barley hulls) on colibacillosis in pigs. However, results have been inconsistent.
Additionally, the typical U.S. diet is very different from European diets. It is thus questionable to directly apply their research findings to our production system. Therefore, our research objectives were: 1) to clarify the impact of different types of dietary fiber, soluble versus insoluble, on colibacillosis; 2) to determine if the addition of an enzyme blend to the high-fiber diets changes the pig’s response to colibacillosis, as enzymes potentially help break down dietary fiber to produce prebiotic oligosaccharides.
We used 60 weaning piglets weighing 6.90 ± 0.07 kilograms. Pigs were randomly allotted to one of six treatments with 10 pigs per treatment:
1) NC: non-challenged negative control,
2) PC: F18-challenged positive control,
3) SF: soluble fiber+F18,
4) IF: insoluble fiber+F18, and
5, 6) SF or IF with enzymes.
The soluble and insoluble fiber sources were 10% sugar beet pulp and 15% low-fat corn distiller’s dried grains with solubles, respectively, and were added at the expense of cornstarch in the control diet. Pigs were orally challenged with F18 ETEC or sham infected with phosphate buffered saline on Day 7 (zero day post inoculation). All pigs were pretested to confirm genetic susceptibility to the F18 strain of E. coli. Pig weights and feed disappearance were recorded to calculate performance. Fecal swabs were collected to evaluate E. coli shedding. Pigs were euthanized on dpi 7 to collect tissues to quantify E. coli attachment in the small intestines.
We found that feeding beet pulp (with and without enzymes) improved post-challenge (dpi 1-7) average daily gain by 53% (Figure 1; 0.26 versus 0.17 kilograms) and tended to improve feed intake. This is quite interesting because the calculated energy intake was comparable between the soluble fiber diet and the positive control. We suspect that pigs fed beet pulp either had a more-developed digestive system and thus content, or they utilized energy more efficiently toward lean growth rather than immune activation.
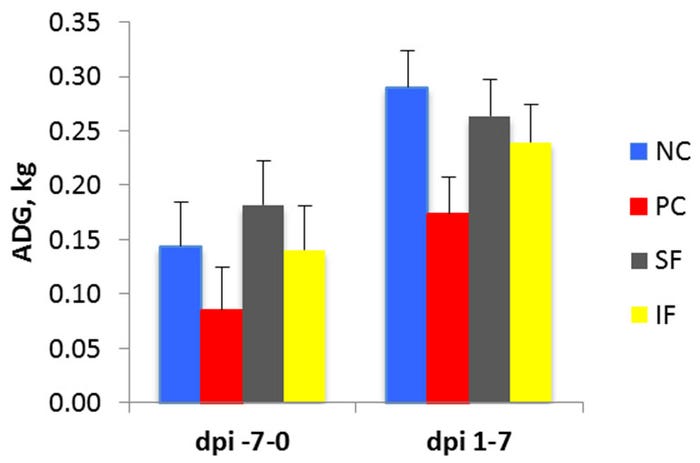
Figure 1: Impact of dietary fiber on average daily gain (kg) in pigs with an F18 E. coli challenge. NC: non-challenge negative control; PC: F18 challenge positive control; SF: combination of soluble fiber with and without enzymes; IF: combination of insoluble fiber with and without enzymes.
Additionally, feeding beet pulp alone without enzymes decreased E. coli attachment in the ileum from 37 to 13% (Figure 2). However, insoluble fiber increased both fecal score and E. coli shedding compared to the positive control, indicating that feeding insoluble fiber from DDGS exacerbated the E. coli infection. This was in agreement with a recent finding by Eric Burrough at Iowa State University who has reported that feeding 30% corn DDGS increased the expression of swine dysentery.
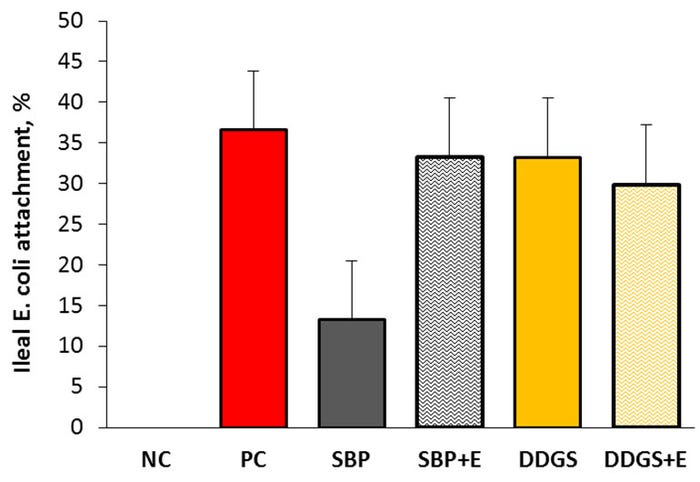
Figure 2: Impact of dietary fiber and enzymes addition on bacterial attachment in the ileum. Results reflect the percent of histologic sections examined with attached coccobacilli morphologically consistent with E. coli.NC: non-challenge negative control; PC: F18 challenge positive control; SBP: soluble fiber; SBP+E: soluble fiber with enzymes; DDGS: insoluble fiber; DDGS+E: insoluble fiber with enzymes.
Our study demonstrated that the addition of 10% highly fermentable soluble fiber mitigated experimentally induced colibacillosis whereas feeding highly insoluble fiber with low fermentability aggravated colibacillosis. Noting that pigs in our study were housed individually under very good hygiene environment as opposed to pigs in production systems, the impact of feeding dietary fiber on E. coli infection may be greater.
In conclusion, our study suggests insoluble fiber from DDGS should be reduced or eliminated in early nursery diets if there is a risk of colibacillosis. The addition of highly fermentable and soluble fiber may be worth consideration when pigs are subject to colibacillosis.
You May Also Like



