September 29, 2014

Porcine epidemic diarrhea – the disease (PED) and the virus that causes it (PEDV) – currently dominate pork producers’ concerns as colder months approach. Detecting evidence for presence of the virus is done by using any of a number of different diagnostic tests. Diagnostic tests can be viewed as “diagnostic tools.” Like a mechanic’s shop, some of the tools are better suited for certain jobs than others, and not every shop has the same repertoire of tools.
There are three general tool types:
Those that detect the agent to answer the question, “Is it there?” For example, to confirm PEDV is present in a clinical specimen, or to try to confirm a negative in a nonclinical surveillance specimen.
Tests that find the agent in a lesion to answer the question, “Is it causing disease in this animal today?”
To determine if animals were previously infected via detection of an antibody and answer the question, “Has this animal been previously infected?”
PCR (polymerase chain reaction) Test
PCR is a common test used to detect segments of nucleic acid (genes) to answer the question, “Is it there?” Depending on application, the design, target and method of a PCR test can be quite different. For example, PCR for PEDV may target one of several different genes (S gene, N gene or M gene), and can be performed as “real-time,” “gel-based,” or “nested.” It is important to recognize that there are differences and underscores the risk in comparing results between laboratories. Laboratories often use different PCR tests and those test results can be interpreted differently depending on sample type, test used and context of sampling. Because PCR only detects a small portion of the PEDV genome, a positive test does not confirm that the sample contains live, viable or infective virus. It is important to note that currently bioassay (feeding some of the sample to susceptible piglets) is about the only way to answer the question “Does this sample contain live, infective PED virus?”
The commonly used “real-time PCR” test results are reported as positive or negative, often in conjunction with a cycle threshold (Ct) number. The “Ct” is the number of times that the PCR machine has to magnify the target (cycles) until it becomes detectable (threshold). Each number increment is a two-fold difference in amount of targeted nucleic acid within the sample. The lower the Ct, the more genomic copies (virus) are present. Each Ct unit is two-fold difference in quantity of virus. For example, there is twice as much target in a sample with Ct of 25 than there is in a sample with Ct of 26. A Ct of 20 is considered quite positive, and a Ct=20 has 1,000x more target in the sample than does a Ct=30. Ct>37 is current considered negative for the target by the Iowa State University Veterinary Diagnostic Laboratory (ISU VDL).
PCR is very sensitive and very specific, perhaps able to detect a single molecule of the target within a test sample. Pigs usually shed PEDV for about 21 days (+/- 10 days) after infection and the virus can be detected in contaminated materials for days, weeks or even months depending on the conditions.
There are two general themes for PCR test application, to confirm PEDV is present when there is disease (“diagnostic mode”) and to attempt to prove PEDV is not present in pigs, environment, feed or substance (“surveillance mode”). These two general applications are at the extremes in the expected amount of virus present in samples. With clinical disease, a large amount of virus is shed whereas in surveillance, one is attempting the impossible – to “prove a negative.”
PCR confirmation of presence of PEDV when there are clinical diarrhea laboratory submissions are straightforward in that very few samples, usually of feces, rectal swabs or oral fluids, are required to be tested to confirm disease. However, when in “surveillance mode,” that is trying to confirm animals or environment (feed, pit, surfaces, transport vehicles, etc.) are negative for virus, many samples of several different sample types may need be tested negative to have confidence that the virus is not there. Consult your veterinarian and/or diagnostic laboratory for appropriate sampling techniques and strategies.
PCR samples should be preserved by immediate refrigeration or freezing. Sample types are many, but most commonly samples will be submitted as tissues, feces, rectal swabs, oral fluids (to confirm clinical disease) as well as environmental wipes, feed, water, manure, etc., surveillance. Please be clean in packaging and shipping samples. Contamination during unpacking of soiled or leaky containers or submission materials can compromise the accuracy of your results as well as results of others.
IHC (immunohistochemistry) and Histopathology
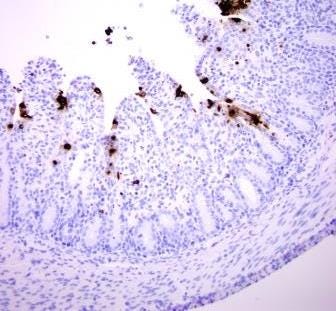
IHC is a useful diagnostic tool which detects antigen within infected cells in formalin-fixed tissues. This technique is often used to answer the question, “Is it important?” The IHC test is much less sensitive in detecting PEDV than is PCR, but it has an advantage in detecting the virus within tissue lesions thereby confirming a role in disease. With PED, the PEDV will be detectable in sections of intestine for five to 10 days (for examples see Figures 1 and 2).
Histopathology and IHC are excellent quality assurance tools that add confidence that the agent(s) detected are actually causing disease. Histopathology can implicate a role for insult types not originally considered, including rotaviruses, E. coli, TGE, coccidia, Salmonella, clostridia or non-infectious contributors. It is important that multiple sections of small intestine (at least six pieces, 1-in. long) from acutely affected pigs be placed in 10% formalin immediately after euthanasia. Be gentle while removing and placing the intestine in formalin as artifacts and distortion can hinder interpretation of lesions and testing.
Figure 1: Early PEDV infection (~8 hrs Post-Infection) in neonatal pig. Infected cells (brown stain) line the villi.
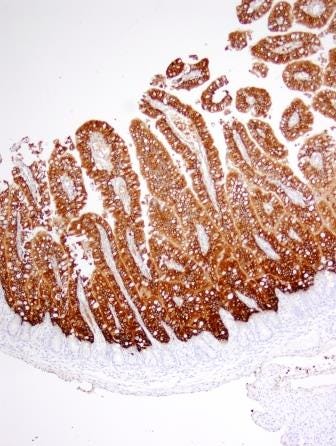
Figure 2: Later PEDV infection (~36 hrs PI): where only a few infected cells remain (brown stain).
Antibody Detection (Serology)
Another set of tools in the diagnostic toolkit are those that detect antibody specific for PEDV. Antibody is formed as part of an immune response and becomes detectable after disease subsides. Antibody testing, sometimes referred to as “serology,” answers the question, “Has it been there?” A positive result suggests that an animal was previously infected (or in young animals, antibody passively acquired via colostrum) but should not be interpreted as predicting animals are “immune” to disease. Similarly, the absence of antibody in serum does not mean there is no immunity, particularly for mucosal pathogens such as PEDV. In general, antibody detectable in serum for PEDV is relatively short-lived, meaning it does not survive three to five months. Samples that contain antibody and may be used for testing are usually serum, but oral fluids, colostrum and milk may also be sometimes used IF the particular test is designed and validated for this.
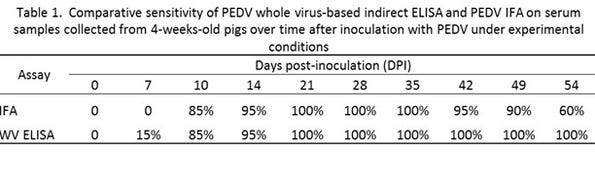
Following the successful isolation and propagation of PEDV in cell culture at the ISU-VDL, an indirect immunofluorescence assay test (IFAT) was developed and implemented in September 2013 for the detection of PEDV antibodies in swine serum. The diagnostic sensitivity and specificity of IFA is estimated to be 95.6% and 98.7%, respectively, using a dilution of ≥ 1:40 as cut-off. However, as noted above, IFAT has been found to have a relatively short duration of positivity (e.g. two months as demonstrated in Table 1). Several ELISA (enzyme-linked immunosorbant assay) tests have been evaluated and are available at various laboratories. Similar to the IFAT, most of the ELISA tests only detect antibody for a relatively short time after infection.
In September 2014, a new whole-virus ELISA test to measure IgG (WV IgG ELISA) became available at ISU-VDL which offers advantages over IFAT, including: amenable to handling large number of samples, rapid turn-around and greater diagnostic sensitivity. The performance of the PEDV WV IgG ELISA was compared to the PEDV IFAT using serum samples collected from four-week-old pigs inoculated with PEDV USA/Iowa/18984/2013 under research conditions (Table 1). Anti-PEDV IgG serum antibody was detected between seven (ELISA) and 10 (IFAT) days post-inoculation (DPI). All animals were positive by DPI 21, but the detection rate and sensitivity of the IFAT declined over time. Thus, the IFAT and ELISA have similar detection rates at early stages of the infection, but the PED WV IgG ELISA test detected antibody longer than the IFAT.
The diagnostic specificity of the PEDV WV ELISA was further evaluated using +500 serum samples collected in December 2011 and January 2012, i.e., these samples were collected when the U.S. swine herd was still free of PEDV. Diagnostic specificity of the ELISA was estimated to be 98.5% in that sample set. Currently, during the onboarding process of this new assay, the ISU VDL is further evaluating the utility of this assay as a PEDV antibody screening assay in serum, oral fluids, milk, colostrum and feces but continues to use the PEDV IFAT as confirmatory test when needed. There is an expectation that the PEDV WV ELISA will be validated for milk, colostrum and oral fluids soon.
You May Also Like



